| Notice: |
This page is derived from the original publication listed below, whose author(s) should always be credited. Further contributors may edit and improve the content of this page and, consequently, need to be credited as well (see page history). Any assessment of factual correctness requires a careful review of the original article as well as of subsequent contributions.
If you are uncertain whether your planned contribution is correct or not, we suggest that you use the associated discussion page instead of editing the page directly.
This page should be cited as follows (rationale):
Fukushima C, Bertani R (2017) Taxonomic revision and cladistic analysis of Avicularia Lamarck, 1818 (Araneae, Theraphosidae, Aviculariinae) with description of three new aviculariine genera01. ZooKeys (659) : 1–185, doi. Versioned wiki page: 2017-03-02, version 136794, https://species-id.net/w/index.php?title=Avicularia&oldid=136794 , contributors (alphabetical order): Pensoft Publishers.
Citation formats to copy and paste
BibTeX:
@article{Fukushima2017ZooKeys,
author = {Fukushima, Caroline Sayuri AND Bertani, Rogério},
journal = {ZooKeys},
publisher = {Pensoft Publishers},
title = {Taxonomic revision and cladistic analysis of Avicularia Lamarck, 1818 (Araneae, Theraphosidae, Aviculariinae) with description of three new aviculariine genera01},
year = {2017},
volume = {},
issue = {659},
pages = {1--185},
doi = {10.3897/zookeys.659.10717},
url = {http://zookeys.pensoft.net/articles.php?id=10717},
note = {Versioned wiki page: 2017-03-02, version 136794, https://species-id.net/w/index.php?title=Avicularia&oldid=136794 , contributors (alphabetical order): Pensoft Publishers.}
}
RIS/ Endnote:
Wikipedia/ Citizendium:
<ref name="Fukushima2017ZooKeys">{{Citation
| author = Fukushima C, Bertani R
| title = Taxonomic revision and cladistic analysis of Avicularia Lamarck, 1818 (Araneae, Theraphosidae, Aviculariinae) with description of three new aviculariine genera01
| journal = ZooKeys
| year = 2017
| volume =
| issue = 659
| pages = 1--185
| pmid =
| publisher = Pensoft Publishers
| doi = 10.3897/zookeys.659.10717
| url = http://zookeys.pensoft.net/articles.php?id=10717
| pmc =
| accessdate = 2025-03-06
}} Versioned wiki page: 2017-03-02, version 136794, https://species-id.net/w/index.php?title=Avicularia&oldid=136794 , contributors (alphabetical order): Pensoft Publishers.</ref>
See also the citation download page at the journal. |
Taxonavigation
Ordo: ORDO
Familia: FAMILIA
Name
Avicularia Lamarck, 1818 – Wikispecies link – Pensoft Profile
- Aranea Linnaeus, 1758: 622 (in part: Avicularia avicularia); Fabricius 1775[1]: 438 (in part: Avicularia avicularia); 1793: 424 (in part: Avicularia avicularia); De Geer 1778[2]: 313, pl. 38, fig. 8 (in part: Avicularia avicularia); 1783: 122, pl. 38, figs 8–10 (in part: Avicularia avicularia); Latreille 1802[3]: 49 (in part: Avicularia avicularia). Name abandoned as generic name to be used for the Order name (Thorell 1870[4]: 151).
- Mygale Latreille, 1802: 49, 1804: 152, pl. LXII, fig.1 (in part: Mygale avicularia); 1806: 85 (in part: Mygale avicularia); Walckenaer 1805[5]: 4 (in part: Mygale avicularia); Hahn 1820[6]: 5 (in part: Mygale avicularia); Simon 1864[7]: 70, figs 26–28 (in part: Mygale avicularia). Name preoccupied in Mammalia by Mygale Cuvier, 1800 (Thorell 1870[4]: 162).
- Avicularia Lamarck, 1818: 107 (type species Aranea avicularia Linnaeus, 1758 by subsequent designation in direction 67 of ICZN (1957[8]: 116)); Thorell 1870[4]: 168; Ausserer 1871[9]: 128, 201; 1875[10]: 138; Simon 1889[11]: 213; 1892[12]: 171; 1903[13]: 958, 960; F. O. Pickard-Cambridge 1896[14]: 740, 746; 1899[15]: 42; Pocock 1901[16]: 548; Mello-Leitão 1923[17]: 320, 376; ICZN 1928[18]: 395; Petrunkevitch 1928[19]: 81; Roewer 1942[20]: 253; Bonnet 1955[21]: 826; Valerio 1979[22]: 307; Raven 1985[23]: 119; Gallon 2008[24]: 243; World Spider Catalog 2016[25].
- Eurypelma C. L. Koch, 1850: 73, 74 (in part); Simon 1864[7]: 67 (in part); Roewer 1942[20]: 238 (in part); Bonnet 1955[21]: 1828; Raven 1985[23]: 146, 153. Considered objective synonym of Avicularia by Raven (1985[23]: 146).
-
-
- Anchylochyrus Petrunkevitch, 1928: 83 (unjustified emendation per Bonnet 1955[21]); Strand 1929[26]: 12.
Type species
Aranea avicularia Linnaeus, 1758, by subsequent designation (ICZN 1957[8]).
Species included
Avicularia avicularia, Avicularia caei sp. n., Avicularia glauca, Avicularia hirschii, Avicularia juruensis, Avicularia lynnae sp. n., Avicularia merianae sp. n., Avicularia minatrix, Avicularia purpurea, Avicularia rufa, Avicularia taunayi and Avicularia variegata stat. n.
Diagnosis
Avicularia resembles Caribena gen. n., Ybyrapora gen. n., Iridopelma and Typhochlaena by the procurve anterior row of eyes (Fig. 13). It can be distinguished from Typhochlaena by the digitiform apical article of PLS (Fig. 10). It differs from Iridopelma by male lacking tibial apopysis on tibiae II and female by spermathecae long, with accentuated outwards curvature medially (Fig. 21). It differs from Caribena gen. n. by stout urticating setae on abdomen dorsum of male and female (Figs 15–17) and by rounded process on cymbium (Fig. 307). From Ybyrapora gen. n. it can be distinguished by sclerotized spermathecae in females (Fig. 21) and presence of developed tibial apophysis in males (Fig. 311), except Avicularia minatrix, Avicularia lynnae sp. n., Avicularia caei sp. n., and A hirschii that lack tibial apophysis. Males of these Avicularia species can be distinguished from Ybyrapora diversipes comb. n. by the embolus less curved in frontal view (Fig. 30). From Ybyrapora sooretama comb. n. and Ybyrapora gamba comb. n. they differ in terms of the presence of a well-developed process on cymbium (Fig. 307), except Avicularia minatrix. Avicularia minatrix can be distinguished from Ybyrapora sooretama comb. n. and Ybyrapora gamba comb. n. by the abdomen dorsum black having orange spots on its side (Fig. 89).
Description
Carapace slightly longer than wide, cephalic region slightly raised. Cephalic and thoracic striae inconspicuous due to setae density. Fovea deep or shallow, slightly recurve (most species) or straight. Chelicera without rastelum. Eye tubercle distinct, raised or slightly raised, wider than long. Anterior row of eyes procurve (Fig. 13), posterior recurve or slightly recurve (most species). Clypeus narrow (most species) or absent. Labium subquadrate, longer than wide, with 50–133 cuspules spaced by one or two diameters from each other on the anterior third center. Maxillary lyra absent. Maxilla subretangular, anterior lobe distinctly produced into conical process, inner angle bearing 85–215 cuspules. Sternum longer than wide, posterior angle acute, not separating coxae IV. Three pairs of sigilla, some pairs sometimes not evident. Anterior ellipsoidal or rounded, middle rounded, posterior ellipsoidal (most species) or rounded. All positioned one diameter or less from margin. Leg formula: I=IV II III (most species) or IV I II III (Avicularia avicularia, Avicularia rufa and Avicularia hirschii female). Clavate trichobothria on distal 2/3 or 1/2 of tarsi I–IV. Tarsi I–IV fully scopulate, IV divided by a band of setae or divided by a bald stripe. Metatarsi I–II fully scopulate in most species, III 1/3 to 1/2 distal scopulate and IV 1/5 to 1/2 distal scopulate. Metatarsi IV divided by a row of setae. Scopulae of tarsi and metatarsi I–II very extended laterally giving them a spatulate appearance (Figs 299–300). Femora IV without retrolateral scopulae. Stridulatory setae absent. Legs aspinose. ITC absent; STC without denticles. Posterior lateral spinnerets digitiform (Fig. 10). Typical stout type II urticating setae on dorsal abdomen in females and males, 0.36 to 0.72 mm long, 0.11–0.019 mm wide in females and 0.78 to 1.11 mm long, 0.013–0.025 mm wide in males (Figs 15–17). Male tibiae I with apophysis (most species) (Fig. 311) or with discrete elevation covered by a cluster of setae on apical portion (Fig. 309), on prolateral side. Tibial apophysis with a single branch having well-developed base and grouped spiniform setae distally (Figs 33–35). Tibiae II lacking apophysis. Male metatarsus I touches retrolaterally tibial apophysis’ setae when folded. Globous bulb with small subtegulum; prominence on prolateral tegulum developed (most species) (Fig. 314), well-developed (Avicularia variegata stat. n. and Avicularia juruensis) (Fig. 315), weakly-developed (Avicularia caei sp. n.) (Fig. 313) or absent (Avicularia minatrix) (Fig. 312). Embolus not flattened, without keels, about 3.0 to 3.5 times tegulum’s length (most species) or more than 4 times tegulum’s length (Avicularia lynnae sp. n. and Avicularia caei sp. n.) in retrolateral view. Embolus medial portion and tegulum’s margin form an acute angle in retrolateral view (Fig. 323). Embolus with basal part very curved in frontal view (Figs 312–315), thin distal width and tip narrowing abruptly (Figs 316–317) or tapering (Figs 318–319). Cymbium subtriangular with subequal lobes, having a well-developed rounded process on retrolateral lobe (most species) (Fig. 307) or lacking any process (Fig. 162). Cymbium process bearing thick setae (most species) (Fig. 307) or thin setae (Fig. 205). Spermathecae with an accentuated outwards curvature medially, completely separated, and long (Fig. 21). Spermathecae not-twisted (except Avicularia hirschii) with walls lacking projections or lobes (except Avicularia taunayi) (Figs 21–27). Spermathecae midwidth as wide as its base width (most species) (Fig. 21) or midwidth expanded, about 1.5 times the apex width (Avicularia variegata stat. n., Avicularia juruensis and Avicularia taunayi) (Fig. 54). Spermathecae with weakly-sclerotized area shorter than half the length of well-sclerotized area (most species) (Fig. 21) or at least same size of well-sclerotized area (Avicularia purpurea and Avicularia merianae sp. n.) (Fig. 154). Abdomen dorsum of females with homogeneously distributed setae (most species) (Fig. 304) or with long guard-setae grouped on lateral and dorsal anterior areas (Figs 302–303). Legs and palps with long guard-setae having homogeneous coloration along its length (Avicularia avicularia, most morphotypes; Avicularia juruensis morphotype 3, Avicularia purpurea, Avicularia taunayi and Avicularia merianae sp. n.) (Fig. 299) or long guard-setae with darker base and contrasting whitish apex (Avicularia avicularia morphotypes 6 and 7; Avicularia variegata stat. n.; Avicularia rufa; and Avicularia juruensis morphotype 2) (Fig. 300). Leg rings on distal femora, tibiae and metatarsi whitish (most species) (Fig. 299) or yellowish (Avicularia avicularia, morphotype 5; Avicularia variegata stat. n., morphotype 2; Avicularia juruensis morphotype 2; and Avicularia rufa) (Fig. 300). All species except Avicularia minatrix showing drastic ontogenetic changes on abdominal color pattern. Brownish juveniles lacking metallic green or blue sheen, with black tarsi contrasting with other lighter articles (except Avicularia purpurea) (Fig. 44). Juveniles having dorsal reddish abdomen with a single central longitudinal stripe and some transversal black stripes on each side (except Avicularia hirschii). Central longitudinal stripe disconnected from transversal stripes (Avicularia avicularia, Avicularia variegata stat. n., Avicularia juruensis and Avicularia rufa) (Fig. 44), connected with all transversal stripes in each side of abdomen (Avicularia purpurea and Avicularia minatrix) (Fig. 167), connected only with the anterior pair of transversal stripes (Avicularia merianae sp. n.), or connected with first and second anterior pair of transversal stripes (Avicularia taunyai) (Fig. 101).
Distribution and habitat
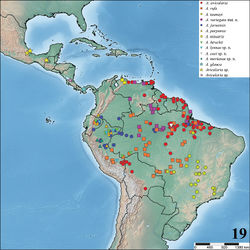
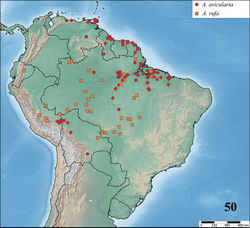
Costa Rica, Panama, Trinidad and Tobago, Venezuela, Guyana, Suriname, French Guiana, Colombia, Ecuador, Peru, Bolivia and Brazil. In Brazil, it occurs in the states of Roraima, Amapá, Amazonas, Pará, Maranhão, Tocantins, Acre, western Bahia, Rondônia, Mato Grosso, Goiás, Minas Gerais, São Paulo, and the Distrito Federal (Fig. 19). Valerio (1979)[22] reports presence of Avicularia species in Costa Rica (see note on Avicularia avicularia redescription and on Avicularia glauca taxonomy). Beside this, there are reports in Lago Catemaco and other localities in the state of Veracruz, as well as in the state of Chiapas, Mexico (R. West and J. Mendoza, pers. comm.), but no Mexican specimens could be examined. Thus, despite the reliable records, it is not possible to know which species are in Mexico.
When erecting the genus Avicularia, Lamarck (1818)[27] included in it three species described originally in Aranea, in this order: Avicularia canceridea (Lamarck, 1818), Avicularia blondii (Latreille, 1804), and Avicularia fasciata (Latreille, 1804). The first species, Avicularia canceridea, could be found in the Antilles in “Meridional America” (South America)(Lamarck 1818[27]). As synonyms of Avicularia canceridea, Lamarck (1818)[27] cited Aranea avicularia Linnaeus, 1758 (sensu Linnaeus 1758[28] and Fabricius 1793[29]) and Mygale avicularia Latreille, 1804 (sensu Latreille 1804[30] and Walckenaer 1805[5]). It is unclear why Lamarck (1818)[27] did not use the pre-existent specific name avicularia instead of canceridea.
Thorell (1870)[4] considered Aranea avicularia Linnaeus “as the type for Avicularia”, and Avicularia canceridea Lamarck and Aranea vestiaria De Geer as its synonyms. Apparently, this was the reason for the Lamarckian combination Avicularia canceridea being buried.
Ausserer (1871)[9] considered Avicularia canceridea Lamarck, Aranea avicularia Linnaeus, Aranea vestiaria De Geer and Mygale avicularia Latreille as synonyms; however, he considered valid the binomy Avicularia vestiaria and stated it as the type species. F. O. Pickard-Cambridge (1899)[15] also considered Avicularia canceridea Lamarck as a synonym of Avicularia avicularia, but stated this last one as the type species. Despite the specific epithet avicularia being the most used for the type species of Avicularia over the years, it was only officially considered as such in 1928 through Opinion 104 (ICZN 1928[18]) and introduced in the official list of specific names in Zoology through Direction 67 of ICZN (ICZN 1957[8]).
Herein, we tried to establish the real identity of Avicularia avicularia as the original description is uninformative and useless for species identification. The type locality, America, is also vague and the existence of types was controversial. Taking into consideration that Aranea avicularia is a very old species described by Linnaeus in the 10th Edition of Systema Naturae (Linnaeus 1758[28]), it would be a difficult task to track a type.
Apparently, Linnaeus (1758)[28] did not designate holotypes. According to Papavero and Llorente-Bousquets (1995)[31], the oldest citation regarding the type concept as we use, nowadays, is dated 1817 by the German dipterologist C. R. W. Wiedmann. In his publication, Wiedmann (1817 apud Papavero and Llorente-Bousquets 1995[31]) approved the idea about designation of types in description of new species and creation of a Normalmuseum where all types should be deposited. Therefore, Linnaeus could not determine types since this concept was elaborated many years after publication of Systema Naturae (Papavero and Llorente-Bousquets 1995[31]).
Linnean holotypes are also a problem for other specialists. Many botanists, for example, argue it is very difficult to know when Linnaeus used a well-defined specimen to describe a plant species. Thus, The Linnean Plant Name Typification Project was created to choose types from the specimens and illustrations that Linnaeus used in arriving at his concept of a species in question (The Linnean Society of London 2016[32]). The creation of this project strengthens the ideia that Linnaeus did not designate holotypes. Specifically in Avicularia’s case, this idea is supported by the expressed citation by F. O. Pickard-Cambridge (1896)[14] that the type is a figure of two specimens drawn by Merian (1726)[33], and not a specimen.
Even with a doubtful existence of the Avicularia avicularia holotype, Gabriel et al. (2007)[34] tried to locate it in The Linnean Society of London Collection. The authors found two specimens of Aranea avicularia considered by the curator as Linnean material (Gabriel et al. 2007[34]) and observed they are two different species: a female of Stromatopelma calceatum Fabricius, 1793; and a male of Harpactira atra Latreille, 1832, both African species (Gabriel et al. 2007[34]). They concluded that none of the African specimens in the Linnean collection could be considered types of Avicularia avicularia (Gabriel et al. 2007[34]).
The ICZN states on the article 72.1.1 of its Code that “in the absence of holotype designation, or the designation of syntypes, or the subsequent designation of a lectotype, all [specimens] are syntypes and collectively they constitute the name-bearing type” (ICZN 1999[35]). Additionally, the article 72.4.1. (ICZN 1999[35]) states that “the type series of a nominal species-group taxon consists of all the specimens included by the author in the new nominal taxon (whether directly or by bibliographic reference)”. Thus, as there is no designation of a specific holotype in the original description, all the specimens used in the publications mentioned by Linnaeus in the description of Aranea avicularia are part of a syntypic series.
In that description, Linnaeus (1758)[28] cited his own previous work (Linnaeus 1749[36]), as well as Clusius (1611)[37], Laet (1633)[38], Piso and Marcgrave (1648)[39], Worm (1655)[40], Olearius (1666)[41] and Merian (1705)[42].
The spider drawn by Clusius (1611)[37] seems, in fact, to be an aviculariine, since it has a hairy body and very developed subungueal tufts (Fig. 2). The spider was illustrated with low cephalic region and could be found in Baya de todos los Sanctos [sic] (Clusius 1611[37]). The animal depicted resembles specimens of Pachistopelma spp., which can be found in Baía de Todos-os-Santos, a bay in the city of Salvador, state of Bahia, northeastern Brazil. Laet (1633)[38] and Piso and Marcgrave (1648)[39] also reproduced this illustration and indicated that this species is very common in Saint Aleixo Island (state of Pernambuco, Brazil), area where Pachistopelma rufoniger specimens are frequent and where also there is no record of Avicularia spp. Other authors (Olearius 1666[41], Worm 1655[40]) used the same Clusius’ spider illustration to refer to this Brazilian species which we found to be a Pachistopelma species.
Merian (1705)[42], on the other hand, illustrated two spiders with very developed and conspicuously colored subungueal tufts, unmistakably Avicularia sp. specimens (Fig. 1). One of them is leaving a typical retreat and feeding on ants and the other specimen is feeding on a small bird. Smith (2000b)[43] stated that, probably because of this illustration, Linnaeus used the specific name “avicularia”, meaning “small bird” in Latin. Merian (1705)[42] had drawn these spiders based on specimens observed during her expedition to Suriname. In the illustration’s comments, there is no indication of locality, nor any information that could lead to an inequivocal identification of the illustrated species.
Linnaeus (1758)[28], in his Aranea avicularia description, also cited two illustrations of Seba (1734)[44] of a spider from Ceylon, now Sri Lanka, which is probably a Poecilotheria specimen, and an unidentified specimen from America, which clearly is not an aviculariine.
Thus, among the syntypic Avicularia avicularia series are specimens of Pachistopelma spp. (Clusius 1611[37], Laet 1633[38], Piso and Margrave 1648, Worm 1655[40], Olearius 1666[41]), Avicularia (Merian 1705[42]) and unidentified specimens (Seba 1704). Apparently, the specimens used and/or illustrated by these authors are lost or have never been deposited in a zoological collection, but only observed in their natural habitat. Smith (2000a)[45] believe that Linnaeus (1758)[28] used the engraving done by Merian (1705)[42] for describing Aranea avicularia, which was considered perfectly acceptable at that time. However, Smith (2000a)[45] did not discard the possibility that Linnaeus had examined specimens collected by Merian, which could be deposited in particular collections, although the author states that much of Merian’s material is lost.
The ICZN Code article 72.4.1.1 states that “for a nominal species or subspecies established before 2000, any evidence, published or unpublished, may be taken into account to determine what specimens constitute the type series” (ICZN 1999[35]). The Code also considered part of the syntypic series specimens of which there is evidence that they were known to the author and recognized by him when the nominal species was established (see example in article 72.4.1.1 of ICZN 1999[35]). After searching some collections, we found three specimens of Aranea avicularia belonging to the Linnean collection at the Museum of Evolution of Uppsala University. Even though there is no irrefutable proof that these specimens once belonged to Linnaeus and were examined by him when describing the species (Dr. Mats Eriksson, pers. comm.), we think that two of three specimens found in the collection were used by Linnaeus in his work. The spiders’ donor was Adolf Fredrik, King of Sweden in the time Linnaeus was writing his Systema Naturae (Kullander 2001[46]). The King had an extensive biological collection composed of plants and animals from all over the world, a common habit of wealthy and significant people of that time (Kullander 2001[46]). During nine weeks between 1751 and 1754, Linnaeus, during that time a professor at Uppsala University, was housed in the Royal Castle to catalogue the royal collection; the result of this work was published in a book (Kullander 2001[46]). It is probable that the King donated some material to Linnaeus, which were brought with him to Uppsala instead of going with most of the King’s material to the Swedish Museum of Natural History in Stockholm. Thus, those specimens of Aranea avicularia could have been really used by Linnaeus (1758)[28] to describe the species and, therefore, they are part of the syntypic series.
King Adolf’s specimens are in a dry, pinned collection, thus they cannot be properly examined and handle (Fig. 20). However, even by photography, it is clear that both are Avicularia avicularia specimens, probable from the morphotype found in the state of Pará, Brazil, since morphological features such as size and coloration match with those seen in specimens found in that area. Therefore, herein, we establish the specimen UUZM 61 from the Uppsala collection as lectotype and the specimen UUZM 62 as paralectotype, solving the oldest and one of the most vexing problems in mygalomorph taxonomy.
Key to Avicularia species
Male of Avicularia glauca and female of Avicularia caei sp.n. and Avicularia lynnae sp. n. are unknown.
Female
Taxon Treatment
- Fukushima, C; Bertani, R; 2017: Taxonomic revision and cladistic analysis of Avicularia Lamarck, 1818 (Araneae, Theraphosidae, Aviculariinae) with description of three new aviculariine genera01 ZooKeys, (659): 1-185. doi
Images
| Figures 1–2. Historical aviculariine drawings. 1 Maria Sybilla Merian’s plate from Metamorphosis insectorum Surinamensium (1705) depicting an Avicularia avicularia eating a bird 2 Clusius’ (1611) [37] drawing of a possible Pachistopelma sp., the oldest known illustration of a tarantula ( Theraphosidae, Aviculariinae) in the New World. |
| Figures 15–18. Urticating setae type II in Aviculariinae. 15 Avicularia juruensis male (CAS 4), showing well-developed barbs along all its length 16 Avicularia variegata stat. n. female (IBSP 7900), showing weakly-developed barbs only near the stalk 17 Avicularia avicularia female morphotype 5 (MNRJ 06916) from Pando, showing developed barbs along almost entire length 18 Caribena versicolor comb. n. male (MNHN−AR 4904), very slender setae with barbs along all length. Scale bars = 0.1 mm. |
| Figure 19. Map showing records of Avicularia Lamarck, 1818 species in Central and South America. |
| Figure 20. Avicularia avicularia (Linnaeus, 1758), types from Uppsala Collection (UUZM). Above, lectotype female (UUZM 61), below paralectotype (UUZM 62). Photo: Mats Eriksson, published with UUZM permission. |
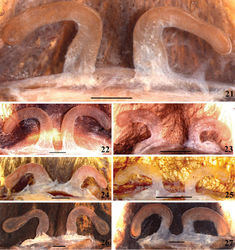
| Figures 21–27. Avicularia avicularia (Linnaeus, 1758), spermathecae variation. 21 morphotype 1, Altamira, state of Pará, Brazil (MNRJ 13995) 22 morphotype 2, UHE Tucuruí, state of Pará, Brazil (IBSP 4885) 23 morphotype 3, Puerto Cabello, state of Carabobo, Venezuela (MNHN–AR 4883) 24 morphotype 4, Cuenca Rio Los Amigos, department of Madre de Dios, Peru (UA 0042/2006) 25 morphotype 5, Yucumo, department of Beni, Bolivia (MNRJ 06919) 26 morphotype 6, Paramaribo, district of Paramaribo, Suriname (AMNH Su59) 27 morphotype 7, Juruti, state of Pará, Brazil (MPEG 15640). Scale bars = 1 mm. |
| Figures 28–35. Avicularia avicularia (Linnaeus, 1758), male (MNRJ 13659A). 28–31 right palpal bulb (mirrored) 28 prolateral 29 retrolateral 30 frontal 31 dorsal 32 right cymbium, dorsal view (mirrored) 33–35 right tibial apophysis of leg I (mirrored) 33 prolateral 34 ventral 35 retrolateral. Scale bars = 1 mm. |
| Figures 36–43. Avicularia avicularia (Linnaeus, 1758) morphotypes. 36–37 morphotype 1 36 female from Castanhal, state of Pará, Brazil 37 immature from Santarém, state of Pará, Brazil 38 morphotype 2, female from Tucuruí, state of Pará, Brazil 39 morphotype 3, female from Caracas, Distrito Capital, Venezuela 40 morphotype 4, female from Tambopata, department of Madre de Dios, Peru 41 morphotype 5, preserved female from Manuripi, department of Pando, Bolivia 42–43 morphotype 6 42 female from Montsinery, department of Cayenne, French Guiana 43 male from Georgetown, department of Demerara-Mahaica, Guyana. Photos: 36 R. Bertani; 37 M. Gamache, 38 W. Bokermann; 39–40, 42–43 R. C. West; 41 C. S. Fukushima. |
| Figures 44–49. Avicularia avicularia (Linnaeus, 1758) morphotypes. 44–46 morphotype 6, from Trinidad and Tobago 44 immature 45 female 46 male 47–49 morphotype 7, from Juruti, state of Pará, Brazil 47 immature 48 female 49 male. Photos: 44–46 R. C. West; 47–49 F. E. Pimenta. |
| Figures 52–53. Avicularia glauca Simon, 1891, holotype female (MNHN–AR 4897). 52 habitus 53 spermathecae. |
| Figures 54–57. Avicularia variegata (F. O. Pickard-Cambridge, 1896) stat. n., spermathecae variation. 54 morphotype 1, Manaus, state of Amazonas, Brazil (INPA 4894) 55 holotype, Itacoatiara, state of Amazonas, Brazil (BMNH 1896.12.13.16) 56 morphotype 2, Alto Alegre, state of Roraima, Brazil (MZUSP 70946) 57 holotype of Avicularia bicegoi Mello-Leitão, 1923, Manaus, state of Amazonas, Brazil (MZUSP 133). Scale bars = 1 mm. |
| Figures 58–65. Avicularia variegata (F. O. Pickard-Cambridge, 1896) stat. n., male (INPA 4897). 58–61 left palpal bulb 58 prolateral 59 retrolateral 60 frontal 61 dorsal 62 left cymbium, dorsal 63–65 left tibial apophysis of leg I 63 prolateral 64 ventral 65 retrolateral. Scale bars = 1 mm. |
| Figures 66–73. Avicularia variegata stat. n., morphotype 1, habitus 66 immature on leaf retreat 67 immature 68 juvenile 69 juvenile female 70 female 71 male 72 adult inside retreat on tree bark 73 female in coconut tree. Photos: 66–69, 71–73 Marlus Almeida; 70 R. C. West. |
| Figures 74–75. Avicularia variegata stat. n., morphotype 2, habitus 74 female 75 male. Photos: 74 C. S. Fukushima; 75 R. Bertani. |
| Figures 77. Map showing records of Avicularia variegata F. O. Pickard-Cambridge, 1896 stat. n. morphotypes. |
| Figures 78–79. Avicularia minatrix Pocock, 1903, spermathecae variation. 78 Venezuela (IBSP 12886) 79 holotype, Duaca, state of Lara, Venezuela (BMNH 1903.7.1.120). Scale bars = 1 mm. |
| Figures 80–87. Avicularia minatrix Pocock, 1903, male (MZUSP 70949). 80–83 right palpal bulb (mirrored) 80 prolateral 81 retrolateral 82 frontal 83 dorsal 84 right cymbium, dorsal (mirrored) 85–87 left tibia I 85 prolateral 86 ventral 87 retrolateral. Scale bars = 1 mm. |
| Figures 88–89. Avicularia minatrix Pocock, 1903, habitus. 88 female, state of Falcón, Venezuela 89 male. Photos: 88 J. Huff; 89 J. Newland. |
| Figures 91–92. Avicularia taunayi (Mello-Leitão, 1920), spermathecae variation. 91 Sobradinho, Brasília, Distrito Federal, Brazil (DZUB 352) 92 Brasíla, Distrito Federal, Brazil (DZUB 1979). Scale bars = 1 mm. |
| Figures 93–100. Avicularia taunayi (Mello-Leitão, 1920), male (DZUB 1675; except cymbium, DZUB 4542). 93–96 left palpal bulb 93 prolateral 94 retrolateral 95 frontal 96 dorsal 97 left cymbium, dorsal 98–100 right tibial apophysis of leg I (mirrored) 98 prolateral 99 ventral 100 retrolateral. Scale bars = 1 mm. |
| Figures 101–104. Avicularia taunayi (Mello-Leitão, 1920), habitus and retreat. 101 immature 102 female 103 male 104 retreat inside trunk of Myrcia tormentosa (Aubl.). Arrow indicating retreat opening. Photos: 101–103 R. Bertani; 104 C. S. Fukushima. |
| Figures 106–108. Avicularia juruensis Mello-Leitão, 1923, spermathecae variation. 106 paralectotype, Rio Juruá, state of Amazonas, Brazil (MZUSP 125C) 107 paralectotype, Rio Juruá, state of Amazonas, Brazil (MZUSP 125A) 108 morphotype 3, Pebas, department of Loreto, Peru (MNHN–AR 4902). Scale bars = 1 mm. |
| Figures 109–116. Avicularia juruensis Mello-Leitão, 1923, male lectotype (MZUSP 125B). 109–112 left palpal bulb 109 prolateral 110 retrolateral 111 frontal 112 dorsal 113 left cymbium, dorsal 114–116 right tibial apophysis of leg I (mirrored) 114 prolateral 115 ventral 116 retrolateral. Scale bars = 1 mm. |
| Figures 117–122. Avicularia juruensis Mello-Leitão, 1923, habitus. 117–118 morphotype 2 117 immature 118 juvenile 119 female with yellowish leg ring, department of Napo, Ecuador 120 female with whitish leg ring, department of Loreto, Peru 121 male, department of Loreto, Peru 122 morphotype 1, female, Peru. Photos: 117 Tanya Stewart; 118 W. Lamar; 119–122 R. C. West. |
| Figures 123–126. Avicularia juruensis Mello-Leitão, 1923, retreat, habitus and behavior. 123 morphotype 2, retreat 124 morphotype 1, female from Cruzeiro do Sul, state of Acre, Brazil, habitus 125 female swimming on Rio Momon, Peru 126 female swimming on Rio Marañon, Peru. Photos: 123, 125–126 R. C. West; 124 S. Albuquerque. |
| Figures 128–130. Avicularia rufa Schiapelli & Gerschman, 1945, spermathecae variation. 128 between vale de São Domingos and Pontes & Lacerda, state of Mato Grosso, Brazil (IBSP 10264) 129 Rio Negro, state of Amazonas, Brazil (AMNH RW29) 130 Tomé-Assu, state of Pará, Brazil (IBSP 3105). Scale bars = 1 mm. |
| Figures 131–138. Avicularia rufa Schiapelli & Gerschman, 1945, male (MCP 13592). 131–134 right palpal bulb (mirrored) 131 prolateral 132 retrolateral 133 frontal 134 dorsal 135 right cymbium, dorsal (mirrored) 136–138 right tibial apophysis of leg I (mirrored) 136 prolateral 137 ventral 138 retrolateral. Scale bars = 1 mm. |
| Figures 139–141. SEM microphotographs of cymbium process in male of Avicularia rufa. 139 dorsal view 140 process, detail, dorsal 141 well-developed setae of process. |
| Figures 142–147. Avicularia rufa Schiapelli & Gerschman, 1945, habitus and retreat. 142 immature 143 juvenile 144 old juvenile 145 female 146 male 147 retreat on bromelid. Photos: 142–145, 147 R. Bertani; 146 W. Bokermann. |
| Figures 148–153. Avicularia rufa Schiapelli & Gerschman, 1945, habitat and retreats. 148 Babaçu palms, a common habitat of Avicularia rufa in state of Rondônia, Brazil 149 retreat of immature on a palm tree 150 detail of retreat 151 retreat over a tree bark 152 use of intern side of babaçu palm to build retreat 153 in human buildings. Photos: 148–152 R. Bertani; 153 C. S. Fukushima. |
| Figures 154–157. Avicularia purpurea Kirk, 1990, spermathecae variation. 154 Puerto Napo, department of Napo, Ecuador (CAS 11) 155 Tena, department of Napo, Ecuador (AMNH RW54) 156 holotype, Tena, department of Napo, Ecuador (BMNH 1990.5.22.1) 157 Iquitos, department of Loreto, Peru (AMNH RW33). Scale bars = 1 mm. |
| Figures 158–165. Avicularia purpurea Kirk, 1990, male (CAS 3; except palpal bulb in prolateral view, CAS 7). 158–161 left palpal bulb 158 prolateral 159 retrolateral 160 frontal 161 dorsal 162 left cymbium, dorsal 163–165 left tibial apophysis of leg I 163 prolateral 164 ventral 165 retrolateral. Scale bars = 1 mm. |
| Figures 166–171. Avicularia purpurea Kirk, 1990, habitus and retreat. 166 silken retreat over tree bark 167 immature 168–169 northern form, from Tena, Ecuador 168 female 169 male 170–171 southern form, from Rio Momon, Peru 170 female 171 male. Photos: 166, 168, 170 R. C. West; 167 R. Bertani; 169 R. Baxter; 171 W. Lamar. |
| Figures 173–180. Avicularia hirschii Bullmer et al. 2006 [47], male holotype (SMF 57125). 173–176 right palpal bulb (mirrored) 173 prolateral 174 retrolateral 175 frontal 176 dorsal 177 right cymbium, dorsal (mirrored) 178–180 left tibia I 178 prolateral 179 ventral 180 retrolateral. Scale bars = 1 mm. |
| Figures 181–183. Avicularia hirschii Bullmer et al. 2006 [47], spermathecae variation. 181 paratype, Oriente, department of Napo, Ecuador (SMF 57126) 182 Estação Ecológica Rio Acre, Assis Brasil, state of Acre, Brazil (MNRJ 06911) 183 Senador Guiomard, state of Acre, Brazil (MNRJ 06912). Scale bars = 1 mm. |
| Figures 184–187. Avicularia hirschii Bullmer et al. 2006 [47], habitus. 184 immature, Tiguino, department of Pastaza, Ecuador 185 juvenile, Senador Guiomard, state of Acre, Brazil 186 female, Estação Ecológica Rio Acre, Assis Brasil, state of Acre, Brazil 187 female feeding on an insect, Rio Momón, department of Loreto, Peru. Photos: 184 W. Lamar; 185 S. Albuquerque, 186 M. A. de Freitas; 187 R. C. West. |
| Figures 188–195. Avicularia merianae sp. n., male holotype (AMNH Pe102). 188–191 left palpal bulb 188 prolateral 189 retrolateral 190 frontal 191 dorsal 192 left cymbium, dorsal 193–195 left tibial apophysis of leg I 193 prolateral 194 ventral 195 retrolateral. Scale bars = 1 mm. |
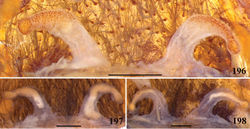
| Figures 196–198. Avicularia merianae sp. n., spermathecae variation. 196 paratype, Moyobamba, department of San Martín, Peru (AMNH Pe102) 197 Tarapoto, department of San Martín, Peru (AMNH Pe106) 198 Moyobamba, department of San Martín, Peru (AMNH Pe100). Scale bars = 1 mm. |
| Figures 199–200. Avicularia merianae sp. n., habitus and retreat. 199 female 200 silken retreat over tree bark. Photos: H.-W. Auer. |
| Figures 201–208. Avicularia lynnae sp. n., male holotype (AMNH RW49). 201–204 left palpal bulb 201 prolateral 202 retrolateral 203 frontal 204 dorsal 205 left cymbium, dorsal 206–208 left tibia I 206 prolateral 207 ventral 208 retrolateral. Scale bars = 1 mm. |
| Figure 209. Avicularia lynnae sp. n., habitus, male from department of Loreto, Peru. Photo: R. C. West. |
| Figures 210–217. Avicularia caei sp. n., male holotype (MPEG 015637). 210–213 right palpal bulb (mirrored) 210 prolateral 211 retrolateral 212 frontal 213 dorsal 214 right cymbium, dorsal (mirrored) 215–217 right tibia I (mirrored) 215 prolateral 216 ventral 217 retrolateral. Scale bars = 1 mm. |
| Figure 218. Avicularia caei sp. n, holotype, habitus, preserved male (MPEG 015637). Detail: lateral abdomen. Arrow indicating stripes. Photo: C. S. Fukushima. |
|
| The maximum number of images (100) is exceeded ! |
Other References
- ↑ Fabricius J (1775) Systema entomologiae, sistens insectorum classes, ordines, genera, species, adiectis, synonymis, locis descriptionibus observationibus. Flensburg and Lipsiae, 832 pp. http://www.museunacional.ufrj.br/mndi/Aracnologia/pdfliteratura/Fabricius%201775%20Systema%20Ent.pdf
- ↑ De Geer C (1778) Mémoires pour servir à l’histoire des insectes. Tome 7, Stockholm, 176–324. https://doi.org/10.5962/bhl.title.14802
- ↑ Latreille P (1802) Histoire naturelle générale et particulière des crustacés et des insectes, ouvrage faisant suite aux oeuvres de Leclerc de Buffon, et partie du cours complet d’histoire naturelle rédigé par C. S. Sonnini/ par P. A. Latreille. Paris, tome 3, 467 pp. https://doi.org/10.5962/bhl.title.15764
- ↑ 4.0 4.1 4.2 4.3 Thorell T (1870) On European spiders. Nova Acta Regiae Societatis Scientiarum Upsaliensis (3) 7: 109–242. https://babel.hathitrust.org/cgi/pt?id=hvd.32044107177446;view=1up;seq=10
- ↑ 5.0 5.1 Walckenaer C (1805) Tableau des aranéides ou caractères essentiels des tribus, genres, familles et races que renferme le genre Aranea de Linné, avec la désignation des espèces comprises dans chacune de ces divisions. Paris, 88 pp. https://doi.org/10.5962/bhl.title.66043
- ↑ Hahn C (1820) Monographie der Spinnen. Nürnberg, Heft 1, 1–16, 4 pls. http://digi.ub.uni-heidelberg.de/diglit/hahn1820/0001
- ↑ 7.0 7.1 Simon E (1864) Histoire naturelle des araignées (aranéides). Paris, 540 pp. https://doi.org/10.5962/bhl.title.47654
- ↑ 8.0 8.1 8.2 ICZN (1957) International Commission on Zoological Nomenclature – Direction 67: Addition to the Official List of Specific Names in Zoology (a) of the specific names of twenty species belonging to the Classes Trilobita, Crustaceae (other than species of the order Decapoda), Diplopoda and Arachnida, each of which is the type species of a genus, the name of which was placed on the Official List of Generic Names in Zoology up to the end of 1936, and (b) of the specific name of species belonging to the Class Arachnida which is currently treated as a senior synonym of the name of such a species. London: The International Trust for Zoological Nomenclature, volume 1, section E, 111–128. http://www.biodiversitylibrary.org/page/34611344#page/133/mode/1up.
- ↑ 9.0 9.1 Ausserer A (1871) Beiträge zur Kenntniss der Arachniden-Familie der Territelariae Thorell (Mygalidae Autor). Verhandlungen der Zoologisch-Botanischen Gesellschaft (Wien) 21: 117–224. http://www.biodiversitylibrary.org/page/16425408#page/271/mode/1up
- ↑ Ausserer A (1875) Zweiter Beitrag zur Kenntniss der Arachniden-Familie der Territelariae Thorell (Mygalidae Autor). Verhandlungen der Zoologisch-Botanischen Gesellschaft (Wien) 25: 125–206. http://www.biodiversitylibrary.org/page/16420158#page/229/mode/1up
- ↑ Simon E (1889) Arachnides. In: Voyage de M. E. Simon au Venezuela (décembre 1887-avril 1888). 4e Mémoire. Annales de la Société Entomologique de France 6(9): 169–220. http://gallica.bnf.fr/ark:/12148/bpt6k6337232k/f163.image
- ↑ Simon E (1892) Histoire naturelle des araignées. Paris, tome 1, 1084 pp. https://doi.org/10.5962/bhl.title.51973
- ↑ Simon E (1903) Histoire naturelle des araignées. Paris, tome 2: 1080 pp. https://doi.org/10.5962/bhl.title.51973
- ↑ 14.0 14.1 Pickard-Cambridge F (1896) On the Theraphosidae of the lower Amazons: being an account of the new genera and species of this group of spiders discovered during the expedition of the steamship “Faraday” up the river Amazons. Proceedings of the Zoological Society of London 1896: 716–766. http://www.biodiversitylibrary.org/page/31200747#page/810/mode/1up
- ↑ 15.0 15.1 Pickard-Cambridge F (1899) Arachnida– Araneida and Opiliones. In: Biologia Centrali-Americana, Zoology. London, volume 2, 41–88. https://doi.org/10.5962/bhl.title.730
- ↑ Pocock R (1901) Some new and old genera of South American Avicularidae. Annals and Magazine of Natural History (7) 8: 540–555. https://doi.org/10.1080/03745480109443359
- ↑ Mello-Leitão C (1923) Theraphosoideas do Brasil. Revista do Museu Paulista 13: 1–438.
- ↑ 18.0 18.1 ICZN (1928) International Commission on Zoological Nomenclature – Opinion 104: 57 Generic names placed in the Official List. Smithsonian Miscellaneous Collections 73(5): 25–28. http://www.biodiversitylibrary.org/page/8911131#page/231/mode/1up
- ↑ Petrunkevitch A (1928) Systema Aranearum. Transactions of the Connecticut Academy of Arts and Sciences 29: 1–270. https://babel.hathitrust.org/cgi/pt?id=coo.31924017862313;view=1up;seq=13
- ↑ 20.0 20.1 Roewer C (1942) Katalog der Araneae von 1758 bis 1940. Bremen 1: 1–1040.
- ↑ 21.0 21.1 21.2 Bonnet P (1955) Bibliographie araneorum. Analyse méthodique de toute la littérature aranéologique jusqu’en 1939. Douladoure, Tolouse, Part 2, tome 2, 919–1926.
- ↑ 22.0 22.1 Valerio C (1979) Arañas terafósidas de Costa Rica (Araneae: Theraphosidae). II. Psalmopoeus reduncus, redescripción, distribución y el problema de dispersión en terafósidas. Revista de Biologia Tropical 27(2): 301–308.
- ↑ 23.0 23.1 23.2 Raven R (1985) The spider infraorder Mygalomorphae (Araneae): Cladistics and systematics. Bulletin of American Museum of Natural History 182: 1–180. http://digitallibrary.amnh.org/handle/2246/955
- ↑ Gallon R (2008) On some poorly known African Harpactirinae, with notes on Avicuscodra arabica Strand, 1908 and Scodra pachypoda Strand, 1908 (Araneae, Theraphosidae). Bulletin of British Arachnological Society 14: 232–246. https://doi.org/10.13156/arac.2008.14.5.232
- ↑ World Spider Catalog (2016). World Spider Catalog. Natural History Museum Bern http://wsc.nmbe.ch, version 17.5
- ↑ Strand E (1929) Zoological and palaeontological nomenclatorical notes. Acta Universitatis Latviensis 20: 1–29.
- ↑ 27.0 27.1 27.2 27.3 Lamarck J (1818) Classe Séptiéme, Les Arachnides, Les Aranéides ou Arachnides Fileuses, 88–108. In: Histoire naturelle des animaux sans vertèbres. Paris, Tome 5, 1–612. https://doi.org/10.5962/bhl.title.12712
- ↑ 28.0 28.1 28.2 28.3 28.4 28.5 28.6 Linnaeus C (1758) Systema naturae per regna tria naturae, secundum classes, ordines, genera, species cum characteribus differentiis, synonymis, locis. Editio decima, reformata. Holmiae, 823 pp. https://doi.org/10.5962/bhl.title.542
- ↑ Fabricius J (1793) Entomologia systematica emendata et aucta, secundum classes, ordines, genera, species, adjectis, synonimis, locis, observationibus, descriptionibus. Volume 2, Hafniae, 519 pp. http://www.museunacional.ufrj.br/mndi/Aracnologia/pdfliteratura/Fabricius%20(1793)%20Entomologia%20systematica.pdf
- ↑ Latreille P (1804) Histoire naturelle générale et particulière des crustacés et des insectes, ouvrage faisant suite aux oeuvres de Leclerc de Buffon, et partie du cours complet d’histoire naturelle rédigé par C. S. Sonnini/ par P. A. Latreille. Paris, tome 7, 413 pp. https://doi.org/10.5962/bhl.title.15764
- ↑ 31.0 31.1 31.2 Papavero N, Llorente-Bousquets J (1995) Principia taxonomica: una introduccion a los fundamentos logicos, filosoficos y metodologicos de las escuelas de taxonomia biologica. Mexico, Universidad Nacional Autonoma de Mexico, vol. 6, 202 pp.
- ↑ Linnean Society of London (2016): The Linnaean Society Plant Name Typification Project https://www.linnean.org/specimen-collections/the-linnaean-plant-typification-project
- ↑ Merian M (1726) Dissertatio de generatione et metamorphosibus insectorum surinamensium. Accedit appendix transformationum piscium in ranas et ranarum in pisces. The Hague.
- ↑ 34.0 34.1 34.2 34.3 Gabriel R, Gallon R, Smith A (2007) Examination of the Avicularia avicularia specimens from the Linnaean Collection. Journal of the British Tarantula Society 23(1): 22–30.
- ↑ 35.0 35.1 35.2 35.3 ICZN (1999) International Code of Zoological Nomenclature (4th edn). The International Trust for Zoological Nomenclature, London. http://www.iczn.org/iczn/index.jsp
- ↑ Linnaeus C (1749) Amoenitates academicae seu Dissertationes variae Physicae, Medicae, Botanicae, antehac seorsim editae nunc collectae et auctae cum tabulis aeneis. Holminae, volume 1, 563 pp. https://doi.org/10.5962/bhl.title.910
- ↑ 37.0 37.1 37.2 37.3 37.4 Clusius C (1611) Curae posteriores, seu Plurimarum non antè cognitarum, aut descriptarum stirpium, peregrinorumque aliquot animalium n. ae descriptiones: Quibus & omnia ipsius Opera, aliaque ab eo versa augentur, aut illustrantur. Leinden, 39 pp. http://bibdigital.rjb.csic.es/ing/Libro.php?Libro=4663
- ↑ 38.0 38.1 38.2 Laet J (1633) Novus Orbis seu descriptionis Indiae Occidentalis Libri XVIII authore Joanne de Laet Antverp. Novis talulis geographicis et variis animantium, Plantarum Fructuumque iconibus illustrata. Leiden. https://doi.org/10.5962/bhl.title.61376
- ↑ 39.0 39.1 Piso W, Marcgrave G (1648) Historia Naturalis Brasiliae, including de medicina Brasiliensis and Historiae rerum naturalium Brasiliae. Elzevier, Leiden, 293 pp. https://doi.org/10.5962/bhl.title.102103
- ↑ 40.0 40.1 40.2 Worm O (1655) Museum Wormianum. Seu historia rerum rariorum, tam naturalium, quam artificialium, tam domesticarum, quam exoticarum, quae Hafniae Danorum in aedibus authoris servantur. Elzevier, Leiden, 434 pp. https://doi.org/10.5962/bhl.title.63844
- ↑ 41.0 41.1 41.2 Olearius A (1666) Gottorffische Kunst-Kammer, worinnen Allerhand ungemeine Sachen, so theils die Natur theils künstliche hände hervor gebracht und bereitet, vor diesem aus allen vier Theilen der Welt zusammen getragen. Schleswig, Germany, 184 pp. https://doi.org/10.5962/bhl.title.60937
- ↑ 42.0 42.1 42.2 42.3 42.4 Merian M (1705) Metamorphosis insectorum surinamensium; ofte verandering der Surinaamsche insecten; Waar in de Surinaamsche rupsen en wormen met alle des zelfs veranderingen na het leven afgebeeld en beschreeven worden, zynde elk geplaast op die gewassen, bloemen en vruchten, daar sy op gevonden zyn; waar in ookde generatie der kikvorschen, wonderbaare padden, hagedissen, slangen, spinnen en mieren werden ver toond en beschreeven, alles in America na het leven en levensgroote geschildert en beschreeven. Selbstverl, Amsterdan, 60 pp. https://doi.org/10.5962/bhl.title.63607
- ↑ Smith A (2000b) The bird eating spiders of 18th and 19th century arachnology, part II. Forum Magazine of the American Tarantula Society 7(2): 23–30.
- ↑ Seba A (1734) Locupletissimi Rerum Naturalium Thesauri Accurata Descriptio Et Iconobus Artificiosissimis Expressio Per Universam Physices Historiam. Wetstenius, Amstelaedami, 178 pp. https://doi.org/10.5962/bhl.title.62760
- ↑ 45.0 45.1 Smith A (2000a) The bird eating spiders of 18th and 19th century arachnology. Forum Magazine of the American Tarantula Society 7(1): 129–134.
- ↑ 46.0 46.1 46.2 Kullander S (2001) Museum Adolphi Friedrich http://linnaeus.nrm.se/zool/welcome.html.en
- ↑ 47.0 47.1 47.2 47.3 Bullmer M, Thierer-Lutz M, Schmidt G (2006) Avicularia hirschii sp. n. (Araneae: Theraphosidae: Aviculariinae), eine neue Vogelspinnenart aus Ekuador. Tarantulas of the World 124: 3–17.
![Figures 1–2. Historical aviculariine drawings. 1 Maria Sybilla Merian’s plate from Metamorphosis insectorum Surinamensium (1705) depicting an Avicularia avicularia eating a bird 2 Clusius’ (1611)[37] drawing of a possible Pachistopelma sp., the oldest known illustration of a tarantula (Theraphosidae, Aviculariinae) in the New World.](https://species-id.net/o/thumb.php?f=Zookeys-659-e10717-g001.jpg&width=250)
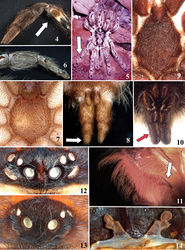
















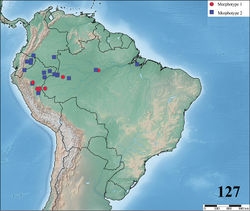
![Figure 90. Map showing records of Avicularia minatrix Pocock, 1903, Avicularia hirschii Bullmer et al. 2006[47], Avicularia lynnae sp. n. and Avicularia caei sp. n.](https://species-id.net/o/thumb.php?f=Zookeys-659-e10717-g023.jpg&width=250)

















![Figures 173–180. Avicularia hirschii Bullmer et al. 2006[47], male holotype (SMF 57125). 173–176 right palpal bulb (mirrored) 173 prolateral 174 retrolateral 175 frontal 176 dorsal 177 right cymbium, dorsal (mirrored) 178–180 left tibia I 178 prolateral 179 ventral 180 retrolateral. Scale bars = 1 mm.](https://species-id.net/o/thumb.php?f=Zookeys-659-e10717-g042.jpg&width=197)
![Figures 181–183. Avicularia hirschii Bullmer et al. 2006[47], spermathecae variation. 181 paratype, Oriente, department of Napo, Ecuador (SMF 57126) 182 Estação Ecológica Rio Acre, Assis Brasil, state of Acre, Brazil (MNRJ 06911) 183 Senador Guiomard, state of Acre, Brazil (MNRJ 06912). Scale bars = 1 mm.](https://species-id.net/o/thumb.php?f=Zookeys-659-e10717-g043.jpg&width=250)
![Figures 184–187. Avicularia hirschii Bullmer et al. 2006[47], habitus. 184 immature, Tiguino, department of Pastaza, Ecuador 185 juvenile, Senador Guiomard, state of Acre, Brazil 186 female, Estação Ecológica Rio Acre, Assis Brasil, state of Acre, Brazil 187 female feeding on an insect, Rio Momón, department of Loreto, Peru. Photos: 184 W. Lamar; 185 S. Albuquerque, 186 M. A. de Freitas; 187 R. C. West.](https://species-id.net/o/thumb.php?f=Zookeys-659-e10717-g044.jpg&width=250)