| Figure 2. Map showing the known distribution of Archaeidae in Australia, with locality records for species of Zephyrarchaea highlighted in black. Note the disjunct distribution of the genus in south-eastern and south-western mainland Australia. |
| Figure 3. Phylogeny of Zephyrarchaea species from Rix and Harvey (2012) [8], showing results from a combined, gene-partitioned Bayesian analysis of that study’s multi-gene dataset (2591 bp: COI, COII, tRNA-K, tRNA-D, ATP8, ATP6, H3). Clades with >95% posterior probability support are denoted by thickened branches, with lower individual clade support values shown above nodes. Note the presence of three main regional clades, each illustrated with an exemplar species (highlighted*). The Victorian species Zephyrarchaea grayi sp. n. and Zephyrarchaea porchi sp. n. were not able to be sequenced for this study (shown as black dots on inset map). |
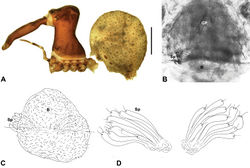
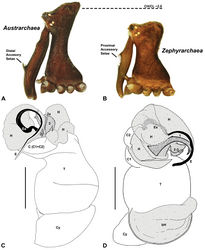
| Figure 4. Diagnostic characters of Zephyrarchaea gen. n. and Austrarchaea Forster & Platnick. A–B, Cephalothorax, lateral view, showing differences in carapace height and the position of accessory setae on male chelicerae: A, male Austrarchaea harmsi Rix & Harvey; B, male Zephyrarchaea marki sp. n. C–D, Expanded male pedipalps, retro-ventral view, showing differences in the articulation and fusion of the conductor sclerites: C, Austrarchaea helenae Rix & Harvey; D, Zephyrarchaea marae sp. n. bH = basal haematodocha; C = conductor; C1–2 = conductor sclerites 1–2; Cy = cymbium; E = embolus; Es = embolic sclerite; H = distal haematodocha; T = tegulum; (TS)1–3 = tegular sclerites 1–3. Scale bars: C–D = 0.2 mm. |
| Figure 5. Carapace morphology of Zephyrarchaea species. A–B, Zephyrarchaea mainae (Platnick): A, male pars cephalica, dorso-lateral view, showing accessory setae (AS) on and adjacent to proximal cheliceral bulge; B, female pars cephalica, antero-lateral view, showing cheliceral foramen (CF) and ocular bulge (OB). C–D, Zephyrarchaea marae sp. n.: C, male cephalothorax, antero-lateral view; D, male pars cephalica and chelicerae, frontal view, showing dorsal ‘head’ region and cheliceral foramen (CF). E, Cheliceraeof male Zephyrarchaea mainae, lateral view, showing proximal accessory setae (AS) and ectal stridulatory file (SF). F–G, Detail of carapace of male Zephyrarchaea marae, lateral view, showing granulate cuticle and setose tubercles (sT). |
| Figure 6. Abdominal morphology of Zephyrarchaea species. A–B, Male abdomens, dorso-lateral view, showing dorsal scutes (S) and additional dorsal sclerites (ds) on hump-like tubercles: A, Zephyrarchaea mainae (Platnick); B, Zephyrarchaea marae sp. n. C, Female epigastric region of Zephyrarchaea mainae, ventral view, showing setose book lung covers (BL) and median genital plate (GP). D, Male epigastric region of Zephyrarchaea mainae, ventral view, showing fusion of epigastric sclerites. E, Abdominal cuticle of male Zephyrarchaea marae, lateral view, showing sclerotic spots (ss) surrounded by short setae. F, Spinnerets of female Zephyrarchaea mainae, posterior view (ventral side uppermost), showing anterior lateral (ALS) and posterior lateral (PLS) spinnerets anterior to anal tubercle (AT), and the absence of a colulus. |
| Figure 7. Graphs depicting the relationship between carapace length (CL) and carapace height (CH) for species of Zephyrarchaea. Smaller grey dots denote species of Austrarchaea (see Rix and Harvey 2011 [8]); boxes denote the three lineages of Zephyrarchaea from Victoria/South Australia, the Stirling Range and elsewhere in southern Western Australia. |
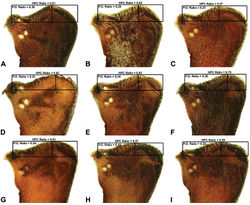
| Figure 9. Lateral ‘head’ profiles of females of species of Zephyrarchaea, showing variation in carapace shape as quantified by the post-ocular ratio (P.O. Ratio) and ratio of highest point of carapace relative to post-ocular length (HPC Ratio) (see Fig. 8): A, allotype Zephyrarchaea vichickmani sp. n.; B, allotype Zephyrarchaea marae sp. n.; C, holotype Zephyrarchaea grayi sp. n.; D, holotype Zephyrarchaea austini sp. n.; E, Zephyrarchaea mainae (Platnick, 1991b); F, allotype Zephyrarchaea janineae sp. n.; G, holotype Zephyrarchaea robinsi (Harvey, 2002a); H, allotype Zephyrarchaea melindae sp. n.; I, allotype Zephyrarchaea barrettae sp. n. |
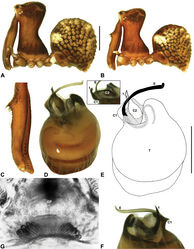
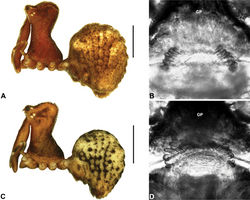
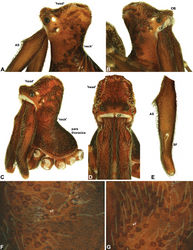
| Figure 10. Zephyrarchaea mainae (Platnick, 1991b). A–B, Cephalothorax and abdomen, lateral view: A, female (WAM T118983) from Mutton Bird Point, Western Australia; B, holotype male (WAM T17683) from Torndirrup National Park, Western Australia. C, Holotype male chelicerae, lateral view, showing accessory setae. D–F, Male (WAM T89569) pedipalp: D–E, bulb, retrolateral view; F, detail of distal tegular sclerites, prolateral view. G, Female (WAM T118983) internal genitalia, dorsal view. C1–2 = conductor sclerites 1–2; E = embolus; GP = genital plate; T = tegulum; (TS)1–3 = tegular sclerites 1–3. Scale bars: A–B = 1.0 mm; E = 0.2 mm. |
| Figure 11. Zephyrarchaea janineae sp. n. A–B, Cephalothorax and abdomen, lateral view: A, allotype female (WAM T118981) from Karri Valley, Western Australia; B, holotype male (WAM T89559) from Karri Valley, Western Australia. C, Holotype male chelicerae, lateral view, showing accessory setae. D–F, Holotype male pedipalp: D–E, bulb, retrolateral view; F, detail of distal tegular sclerites, prolateral view. G, Allotype female internal genitalia, dorsal view. C1–2 = conductor sclerites 1–2; E = embolus; GP = genital plate; T = tegulum; (TS)1–3 = tegular sclerites 1–3. Scale bars: A–B = 1.0 mm; E = 0.2 mm. |
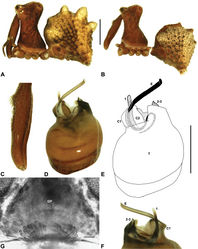
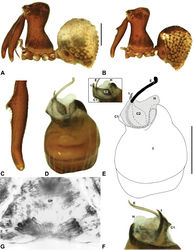
| Figure 12. Zephyrarchaea marki sp. n. A–E, Holotype male (WAM T118985) from Thistle Cove, Cape Le Grand National Park, Western Australia: A, cephalothorax and abdomen, lateral view; B, chelicerae, lateral view, showing accessory setae; C–D, pedipalpal bulb, retrolateral view; E, detail of distal tegular sclerites, prolateral view. C1–2 = conductor sclerites 1–2; E = embolus; T = tegulum; (TS)1–3 = tegular sclerites 1–3. Scale bars: A = 1.0 mm; D = 0.2 mm. |
| Figure 13. Zephyrarchaea robinsi (Harvey, 2002a). A–D, Holotype female (WAM T42580) from Ellen Peak, Stirling Range National Park, Western Australia: A, cephalothorax and abdomen, lateral view; B–D, internal genitalia, dorsal view; C–D, illustrations (from Harvey 2002a) of internal genitalia, dorsal view, showing shape of membranous bursa and underlying spermathecae. B = bursa; GP = genital plate; Sp = spermathecae. Scale bar: A = 1.0 mm. |
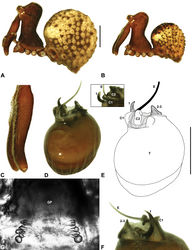
| Figure 14. Zephyrarchaea melindae sp. n. A–B, Cephalothorax and abdomen, lateral view: A, allotype female (WAM T97468) from Toolbrunup Peak, Stirling Range National Park, Western Australia; B, holotype male (WAM T118986) from Mount Hassell, Stirling Range National Park, Western Australia. C, Holotype male chelicerae, lateral view, showing accessory setae. D–F, Holotype male pedipalp: D–E, bulb, retrolateral view, with inset showing twisted apex of tegular sclerite 1 in retroventral view; F, detail of distal tegular sclerites, prolateral view. G, Allotype female internal genitalia, dorsal view. C1–2 = conductor sclerites 1–2; E = embolus; GP = genital plate; T = tegulum; (TS)1 = tegular sclerite 1. Scale bars: A–B = 1.0 mm; E = 0.2 mm. |
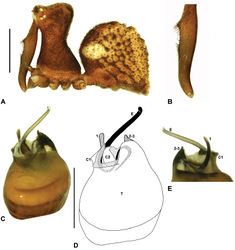
| Figure 15. Zephyrarchaea barrettae sp. n. A–B, Cephalothorax and abdomen, lateral view: A, allotype female (WAM T97466) from Talyuberlup Peak, Stirling Range National Park, Western Australia; B, holotype male (WAM T117055) from Talyuberlup Peak, Stirling Range National Park, Western Australia. C, Holotype male chelicerae, lateral view, showing accessory setae. D–F, Holotype male pedipalp: D–E, bulb, retrolateral view, with inset showing twisted apex of tegular sclerite 1 in retroventral view; F, detail of distal tegular sclerites, prolateral view. G, Allotype female internal genitalia, dorsal view. C1–2 = conductor sclerites 1–2; E = embolus; GP = genital plate; H = distal haematodocha; T = tegulum; (TS)1 = tegular sclerite 1. Scale bars: A–B = 1.0 mm; E = 0.2 mm. |
| Figure 16. Zephyrarchaea vichickmani sp. n. A–B, Cephalothorax and abdomen, lateral view: A, allotype female (MV K11579) from Acheron Gap, Victoria; B, holotype male (MV K11578) from Acheron Gap, Victoria. C, Holotype male chelicerae, lateral view, showing accessory setae. D–F, Holotype male pedipalp: D–E, bulb, retrolateral view, with inset showing twisted apex of tegular sclerite 1 in retroventral view; F, detail of distal tegular sclerites, prolateral view. G, Allotype female internal genitalia, dorsal view. C1–2 = conductor sclerites 1–2; E = embolus; GP = genital plate; T = tegulum; (TS)1–3 = tegular sclerites 1–3. Scale bars: A–B = 1.0 mm; E = 0.2 mm. |
| Figure 17. Zephyrarchaea marae sp. n. A–B, Cephalothorax and abdomen, lateral view: A, allotype female (MV K5921) from Gunyah Rainforest State Reserve, Victoria; B, holotype male (MV K11580) from Tarra-Bulga National Park, Victoria. C, Holotype male chelicerae, lateral view, showing accessory setae. D–F, Holotype male pedipalp: D–E, bulb, retrolateral view, with inset showing twisted apex of tegular sclerite 1 in retroventral view; F, detail of distal tegular sclerites, prolateral view. G, Allotype female internal genitalia, dorsal view. C1–2 = conductor sclerites 1–2; E = embolus; GP = genital plate; T = tegulum; (TS)1–3 = tegular sclerites 1–3. Scale bars: A–B = 1.0 mm; E = 0.2 mm. |
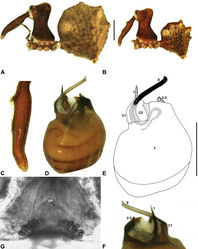
| Figure 18. Zephyrarchaea porchi sp. n. A–E, Holotype male (MV K11581) from Bimbi Park, Otway Range, Victoria: A, cephalothorax and abdomen, lateral view; B, chelicerae, lateral view, showing accessory setae; C–D, pedipalpal bulb (partially expanded), retrolateral view; E, detail of distal tegular sclerites, prolateral view. C1–2 = conductor sclerites 1–2; E = embolus; eH = embolic (distal) haematodocha; T = tegulum; (TS)1–3 = tegular sclerites 1–3. Scale bars: A = 1.0 mm; D = 0.2 mm. |
| Figure 19. Zephyrarchaea grayi sp. n. and Zephyrarchaea austini sp. n. A–B, holotype female Zephyrarchaea grayi sp. n. (AMS KS109448) from Delley’s Dell, Grampians National Park, Victoria: A, cephalothorax and abdomen, lateral view; B, internal genitalia, antero-dorsal view. C–D, holotype female Zephyrarchaea austini (SAM NN28000) from Western River Wilderness Protection Area, Kangaroo Island, South Australia: C, cephalothorax and abdomen, lateral view; D, internal genitalia, antero-dorsal view. GP = genital plate. Scale bars: A, C = 1.0 mm. |
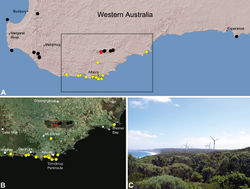
| Figure 20. Zephyrarchaea mainae (Platnick, 1991b), distribution and habitat: A, topographic map showing the known distribution of Archaeidae in south-western Western Australia, with collection localities for Zephyrarchaea mainae highlighted in yellow (red highlighted localities denote juvenile specimens of tentative identification; see Zephyrarchaea sp. unidentified juvenile specimens, above); B, satellite image showing detail of inset (A); C, temperate coastal heathland near the type locality – Albany Wind Farm, Torndirrup Peninsula, Western Australia (March 2008). Image (C) by M. Rix. |
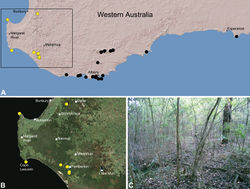
| Figure 21. Zephyrarchaea janineae sp. n., distribution and habitat: A, topographic map showing the known distribution of Archaeidae in south-western Western Australia, with collection localities for Zephyrarchaea janineae highlighted in yellow; B, satellite image showing detail of inset (A); C, wet eucalypt forest at the type locality – Karri Valley, Western Australia (August 2006). Image (C) by M. Rix. |
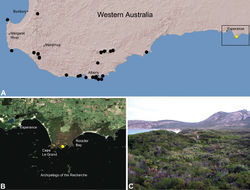
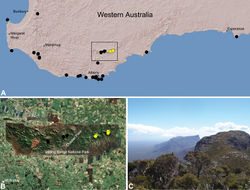
| Figure 22. Zephyrarchaea marki sp. n., distribution and habitat: A, topographic map showing the known distribution of Archaeidae in south-western Western Australia, with collection localities for Zephyrarchaea marki highlighted in yellow; B, satellite image showing detail of inset (A); C, temperate coastal heathland at the type locality – Thistle Cove, Cape Le Grand National Park, Western Australia (June 2010). Image (C) by M. Rix. |
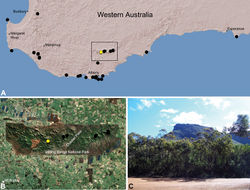
| Figure 23. Zephyrarchaea robinsi (Harvey, 2002a), distribution and habitat: A, topographic map showing the known distribution of Archaeidae in south-western Western Australia, with collection localities for Zephyrarchaea robinsi highlighted in yellow; B, satellite image showing detail of inset (A); C, montane heathland at the type locality – Ellen Peak, Stirling Range National Park, Western Australia (November 2007), with Bluff Knoll visible in the distance. Image (C) by M. Rix. |
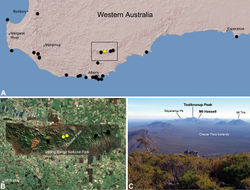
| Figure 24. Zephyrarchaea melindae sp. n., distribution and habitat: A, topographic map showing the known distribution of Archaeidae in south-western Western Australia, with collection localities for Zephyrarchaea melindae highlighted in yellow; B, satellite image showing detail of inset (A); C, view from the summit of Bluff Knoll across the western Stirling Range National Park, showing collection localities for Zephyrarchaea melindae (i.e. Toolbrunup Peak, Mount Hassell) highlighted in bold (June 2010). Note the Chester Pass lowlands, separating populations of Zephyrarchaea melindae and Zephyrarchaea robinsi, and Talyuberlup Peak in the distance, home to Zephyrarchaea barrettae sp. n. Image (C) by M. Rix. |
| Figure 25. Zephyrarchaea barrettae sp. n., distribution and habitat: A, topographic map showing the known distribution of Archaeidae in south-western Western Australia, with collection localities for Zephyrarchaea barrettae highlighted in yellow; B, satellite image showing detail of inset (A); C, view from Stirling Range Drive showing the type locality – Talyuberlup Peak, Stirling Range National Park (August 2008). Image (C) by M. Rix. |
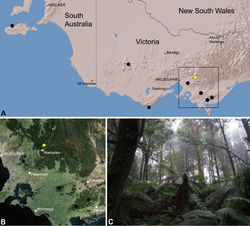
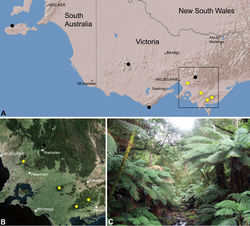
| Figure 26. Zephyrarchaea vichickmani sp. n., distribution and habitat: A, topographic map showing the known distribution of Archaeidae in Victoria and South Australia, with collection localities for Zephyrarchaea vichickmani highlighted in yellow; B, satellite image showing detail of inset (A); C, cool-temperate Nothofagus rainforest at the type locality – Acheron Gap, Yarra Ranges National Park, Victoria (March 2010). Image (C) by M. Rix. |
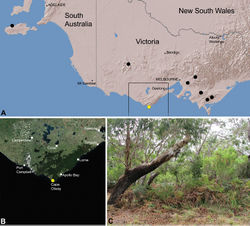
| Figure 27. Zephyrarchaea marae sp. n., distribution and habitat: A, topographic map showing the known distribution of Archaeidae in Victoria and South Australia, with collection localities for Zephyrarchaea marae highlighted in yellow; B, satellite image showing detail of inset (A); C, cool-temperate Nothofagus rainforest at the type locality – Tarra Valley, Tarra-Bulga National Park, Victoria (April 2010). Image (C) by M. Rix. |
| Figure 28. Zephyrarchaea porchi sp. n., distribution and habitat: A, topographic map showing the known distribution of Archaeidae in Victoria and South Australia, with collection localities for Zephyrarchaea porchi highlighted in yellow; B, satellite image showing detail of inset (A); C, bracken-rich eucalypt forest at the type locality – Bimbi Park, Otway Range, Victoria (March 2012). Image (C) by N. Porch, used with permission. |
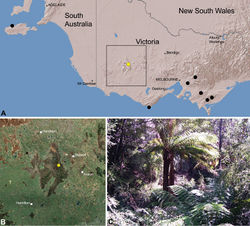
| Figure 29. Zephyrarchaea grayi sp. n., distribution and habitat: A, topographic map showing the known distribution of Archaeidae in Victoria and South Australia, with collection localities for Zephyrarchaea grayi highlighted in yellow; B, satellite image showing detail of inset (A); C, wet sclerophyll forest at the type locality – Delley’s Dell, Grampians National Park, Victoria (March 2010). Image (C) by M. Rix. |
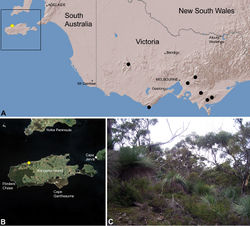
| Figure 30. Zephyrarchaea austini sp. n., distribution and habitat: A, topographic map showing the known distribution of Archaeidae in Victoria and South Australia, with collection localities for Zephyrarchaea austini highlighted in yellow; B, satellite image showing detail of inset (A); C, open eucalypt woodland and heathland at the type locality – near Billy Goat Falls, Western River Wilderness Protection Area, Kangaroo Island, South Australia (May 2010). Image ( C) by M. Rix. |
|

![Figure 3. Phylogeny of Zephyrarchaea species from Rix and Harvey (2012)[8], showing results from a combined, gene-partitioned Bayesian analysis of that study’s multi-gene dataset (2591 bp: COI, COII, tRNA-K, tRNA-D, ATP8, ATP6, H3). Clades with >95% posterior probability support are denoted by thickened branches, with lower individual clade support values shown above nodes. Note the presence of three main regional clades, each illustrated with an exemplar species (highlighted*). The Victorian species Zephyrarchaea grayi sp. n. and Zephyrarchaea porchi sp. n. were not able to be sequenced for this study (shown as black dots on inset map).](https://species-id.net/o/thumb.php?f=ZooKeys-191-001-g003.jpg&width=250)

![Figure 7. Graphs depicting the relationship between carapace length (CL) and carapace height (CH) for species of Zephyrarchaea. Smaller grey dots denote species of Austrarchaea (see Rix and Harvey 2011[8]); boxes denote the three lineages of Zephyrarchaea from Victoria/South Australia, the Stirling Range and elsewhere in southern Western Australia.](https://species-id.net/o/thumb.php?f=ZooKeys-191-001-g007.jpg&width=202)