Trechus bouilloni
| Notice: | This page is derived from the original publication listed below, whose author(s) should always be credited. Further contributors may edit and improve the content of this page and, consequently, need to be credited as well (see page history). Any assessment of factual correctness requires a careful review of the original article as well as of subsequent contributions.
If you are uncertain whether your planned contribution is correct or not, we suggest that you use the associated discussion page instead of editing the page directly. This page should be cited as follows (rationale):
Citation formats to copy and paste
BibTeX: @article{Faille2012ZooKeys217, RIS/ Endnote: TY - JOUR Wikipedia/ Citizendium: <ref name="Faille2012ZooKeys217">{{Citation See also the citation download page at the journal. |
Ordo: Coleoptera
Familia: Carabidae
Genus: Trechus
Name
Trechus bouilloni Faille, Bourdeau & Fresneda sp. n. – Wikispecies link – ZooBank link – Pensoft Profile
Type locality
Spain, Navarra,Sierra de Urbasa–Andía, Lizarraga, Puerto de Lizarraga, UTM (WGS 84): 30 T, X: 580, Y: 4746, Z: 900 m.
Type series
Holotype (MNHN): 1 ♂, Spain, Navarra,Sierra de Urbasa–Andía, Lizarraga, Puerto de Lizarraga, MSS, trap: 1–5–1980/15–8–1980, Bourdeau and Fresneda leg., voucher number ZSM–L201, MNHN]. DNA aliquotes preserved in the DNA and tissue collections of the ZSM, MNHN and IBE; Genitalia dissected and mounted in a separate label pinned with the specimen. Paratypes: 52 ♂♂, 62 ♀♀, same label data as holotype (MNCN, MNHN, MZB, ZSM, CCB, CJF, CAF, CMT).
Diagnosis
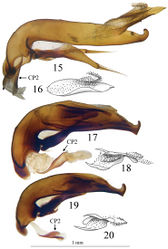
Large size (ca 5 mm) and round shape (Fig. 1). Median lobe of aedeagus slender, in lateral view (Fig. 15) the basal third curved, the central part straight and the apex with a curved hook assymetrical in dorsal view (Fig. 8). Inner sac of aedeagus (=endophallus) with an elongate and well-sclerotized piece, forming a gut and armed with internal scales. Characteristic secondary sclerotization of the sperm duct (Fig. 15: CP2) forming a kind of second copulatory piece outside base of the median lobe.
Description of the holotype
Habitus as in Fig. 1.Elongated, round–sided. Body surface with a very thin, hardly visible, dense microreticulation, with more distinguishable meshes on the head.
Colour. Dorsal surface dark brown, moderately shiny. Antennae, palpi and legs light brown.
Chetotaxy. Surface of elytra glabrous with the exception of a periscutellar seta, two discal setae on the third stria, four humeral setae, four setae along lateral margin and two preapical setae. Marginal setae of pronotum present, the anterior ones located before the first third of the length. Ventral pubescence limited to one seta on each half sternite.
Head. Eyes reduced, flat; ommatidia well defined; maximum diameter of about eight ommatidia, temples approximately twice the length of eyes, strongly wrinkled to the neck. Frontal furrows deeply impressed. Antennae moderately long, five antennomeres extend beyond the pronotal base. Antennomere III distinctly longer than antennomeres II and IV, which are similar in length.
Pronotum. Proportions (M–F): WP/LP = 1.3–1.28, WP/WPB = 1.3–1.3, WP/WH = 1.38–1.3, WE/WP = 1.57–1.53. Transverse, with lateral margins finely bordered; wider in anterior part, narrower than elytra; posterior part much narrower than base of elytra. One seta in the marginal gutter at about a third of pronotum length, another one close to hind angle. Sides evenly rounded and straight just between hind angles and insertions of posterior setae. Hind angles well developed, salient.
Elytra. Proportions (M–F): WE/LE = 0.65–0.69. Oval, broadest almost at mid–length; surface moderately convex, flattened on disc. Shoulders distinct but rounded. Striae very finely punctuated, sixth inner striae deeply impressed on disc, but reduced at apex and base; seventh striae shallower, but distinct, the eighth reduced to the posterior quarter of elytra. Apical striola strongly impressed continuing the fifth stria.
Hind wings. Very reduced, not functional.
Male genitalia. Median lobe of aedeagus slender, in lateral view (Fig. 15) the basal third curved, the central part straight and the apex showing a curved hook; assymetrical in dorsal view (Fig. 8). Parameres slender, each with 4 to 6 setae at apex. Internal sac of aedeagus with an elongate well-sclerotized piece, forming a symmetrical gut and armed with internal scales (Fig. 16). Characteristic secondary sclerotization of the sperm duct forming a kind of second copulatory piece out of the base of the median lobe (Fig. 15: CP2).
Female genitalia
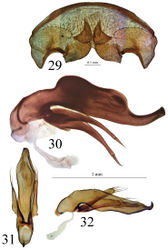
Internal genitalia membranous. Gonocoxites unguiform, with 4 to 5 large setae, and 2 small near apex. Gonosubcoxites with 2 to 3 large setae near the internal edge. Laterotergite IX with 12 setae at the basal margin, and 4 to 6 scattered (Fig. 29).
Size
Mean length (5 exemplars): 5.25 mm (male), 4.56 mm (female).
Etymology
The new species is dedicated to Michel Bouillon, Pyrenean speleologist, who was the first to discover the existence of cave beetles in MSS.
Affinities
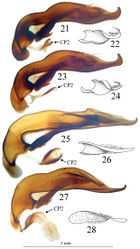
Trechus bouilloni sp. n. is a representative of the Trechus brucki group sensu novo as defined in the present paper. It shares with Trechus grenieri, Trechus uhagoni, Trechus beusti, and Trechus pieltaini the same kind of aedeagus morphology, especially the apex with a curved hook in lateral view, and an internal sac showing two sclerotized parts, the internal copulatory piece and another triangular piece forming a kind of second copulatory piece (CP2, Figs 17–24), also existing in Trechus brucki and Trechus bruckoides sp. n. (Figs 25–28). Similar secondary sclerotized structures of endophallus are known in some groups of insects including Coleoptera, and described as a “sperm pump” (Beutel and Leschen 2005[1], Hünefeld and Beutel 2005[2], Jäch and Delgado 2010[3]). In the Trechus brucki group, however, the structure is too rudimentary to play the same role in sperm transfer, and its function –if any– remains obscure. Although never observed before, this structure is also present in the others species of the group and is the main synapomorphy of the clade. The lack of this internal sclerotized structure in Trechus carrilloi and Trechus sharpi (Figs 30–32) casts doubt on their affinities.
Distribution and ecology
Trechus bouilloni sp. n. is only known from the type locality, the MSS of Lizarraga pass (Navarra, Spain) (Fig. 36). The type locality is a MSS located on a northern slope at the eastern extremity of the Sierra de Andía–Urbasa, close to the Lizarraga pass.
Trechus were collected by means of traps in a zone of scree (altitude: 900 m) extending from east to west at the feet of cliffs of Albian limestone lining the northern slope of the plateau of the Sierra de Andía–Urbasa. This scree slope consists of a mass of fallen rocks resulting from the erosion of calcareous cliffs and constitutes a steeply sloped (45°) MSS, filling one of the numerous gullies of a beech forest covering the entire northern side of the plateau lining the southward depression of the Río Arakil (Sakana valley).
On this unstable ground, beeches are replaced by grassy and mossy vegetation dotted with shrubs. The layer of humus is irregular and very thin and only partly covers the blocks of white, angular, medium–sized limestone, rarely exceeding the size of 1 dm³.
The traps were placed 50 centimeters deep in a “C–type” horizon (sensu Juberthie et al. 1981[4]), constituted mainly by stones of 5 cm³, not sealed by the ground and not welded, leaving numerous spaces between them and forming a layer several meters thick above the compact rock.
The other Coleoptera collected with Trechus bouilloni sp. n. were Leiodidae, Cholevinae: Catops subfuscus Kellner, 1846, Sciodrepoides watsoni (Spence, 1813) (Catopini) and Bathysciola sp. (Leptodirini).
Some specimens of Trechus bouilloni sp. n. were parasitized by an undetermined Ascomycete.
Trechus bouilloni sp. n. was not found in caves of the area north of Larraona (cueva de los Cristinos, cuevas de Erbeltz, Txintxoleze, Noriturri, Akuandi, del Queso, Iniriturri, Arleze, Laminatitur), suggesting that it is strictly located in MSS (CB personal observation).
Original Description
- Faille, A; Bourdeau, C; Fresneda, J; 2012: Molecular phylogeny of the Trechus brucki group, with description of two new species from the Pyreneo-Cantabrian area (France, Spain) (Coleoptera, Carabidae, Trechinae) ZooKeys, 217: 11-51. doi
Other References
- ↑ Beutel R, Leschen R (2005) Phylogenetic analysis of Staphyliniformia (Coleoptera) based on characters of larvae and adults. Systematic Entomology 30: 510-548. doi: 10.1111/j.1365-3113.2005.00293.x
- ↑ Hünefeld F, Beutel R (2005) The sperm pumps of Strepsiptera and Antliophora (Hexapoda). Journal of Zoological Systematics and Evolutionary Research 43: 297-306. doi: 10.1111/j.1439-0469.2005.00327.x
- ↑ Jäch M, Delgado J (2010) Order Coleoptera, family Hydraenidae. In: van Harten, A. (Ed.), Arthropod fauna of the UAE, Vol. 3, Dar Al Ummah Publications, Abu Dabhi, 173–194.
- ↑ Juberthie C, Bouillon M, Delay B (1981) Sur l’existence du milieu souterrain superficiel en zone calcaire. Mémoires de Biospéologie 8: 77-93.
Images
|