Neoneurus vesculus
| Notice: | This page is derived from the original publication listed below, whose author(s) should always be credited. Further contributors may edit and improve the content of this page and, consequently, need to be credited as well (see page history). Any assessment of factual correctness requires a careful review of the original article as well as of subsequent contributions.
If you are uncertain whether your planned contribution is correct or not, we suggest that you use the associated discussion page instead of editing the page directly. This page should be cited as follows (rationale):
Citation formats to copy and paste
BibTeX: @article{Gómez2011ZooKeys125, RIS/ Endnote: TY - JOUR Wikipedia/ Citizendium: <ref name="Gómez2011ZooKeys125">{{Citation See also the citation download page at the journal. |
Ordo: Hymenoptera
Familia: Braconidae
Genus: Neoneurus
Name
Neoneurus vesculus van Achterberg & Gómez sp. n. – Wikispecies link – ZooBank link – Pensoft Profile
Type material
Holotype, ♀ (RMNH),“Spain, Madrid, Carretera de La Coruña km 7.5, 20.viii.2010, following adult workers of Formica cunicularia, J.M. Gómez Durán, RMNH”. Paratypes: 11 ♀ (RMNH (8), RMSEL (2), RMS (1)), topotypic, collected 3.ix. 2010, 13.ix.2010, 24.vi.2011 and 27.vi.2011.
Oviposition behaviour
In recent years the oviposition behaviour of the genus Neoneurus was studied for the first time (Shaw 1992[1], 1993[2]) with Neoneurus mantis Shaw, 1992. Shaw proposed a “raptorial hypothesis” to explain the greatly modified morphology of Neoneurus fore legs (compression of the fore femur, robustness and shortening of the fore tibia, enlargement of the tibial spur, development of a tibial carina often associated with sharp tubercles and spines, shortening of the tarsus and enlargement of the fore pulvillus). These features, together with the two peculiar spinules situated in the frontal area of the head, could serve to grasp the ant before oviposition. Here we confirm for Neoneurus vesculus sp. n. Shaw’s raptorial hypothesis. While this author mentions the metasoma of Formica pozdzolica as the location for alighting and oviposition for Neoneurus mantis, our observations show that Neoneurus vesculus sp. n. alights and probably oviposits in the mesosoma of Formica cunicularia Latreille, 1789.
The observations were made in Madrid (at the enclosed area of the Institute for Agriculture and Food Research and Technology (INIA), Carretera de La Coruña Km 7.5, Spain) during August and September, 2010, in three colonies of Formica cunicularia situated in the base of Atlas cedar trees (Cedrus atlantica). Neoneurus vesculus sp. n. could be seen around the nest entrances in the morning and in the afternoon, with a peak activity of oviposition attacks between 4–7 PM. Two strategies were observed (Fig. 62): a) the perching behaviour as described by Shaw (1993)[2], the wasp standing on a grass stem, on a tree trunk (in both cases at a height less than 5 cm), or on the ground, until an ant approaches; a moment later the wasp takes flight and begins its attack (Movie Neoneurus, first sequence, Appendix III); b) the hovering behaviour -at a distance of about 1 cm- over ants leaving the nest entrance and going up the tree trunk at a height of 3 or 4 cm from the ground. Oviposition attacks following hovering behaviour (Movie Neoneurus, second sequence) increased in the afternoon, being then predominant over the perching behaviour.
When the ant moves up, the wasp approaches it from behind and waits until the ant’s body is in a vertical position. Then, the wasp head hits the ant’s mesonotum while the fore legs dart forward and brace the mesopleuron. The frame analysis reveals that the tibia are the part of the legs that firmly hold the mesopleuron (Figs 63-65).
After contact, the wasp’s head is separated from the ant’s body, the wasp’s metasoma is placed vertically and its wings are folded. Then ovipositor insertion begins, during which time the middle legs can be seen to be sometimes holding the posterior part of the ant’s mesosoma (Fig. 64). The wasp’s metasoma is bent towards the posterior lower part of the ant’s mesosoma, going between the metasoma and the hind leg of the ant. This occurred in a surprisingly asymmetric fashion: of 29 ovipositions observed, the wasp always bent its metasoma between the left hind leg of the ant and the left side of its metasoma. This suggests some asymmetrical morphology of the ovipositor system. According to the frame analysis, the ovipositor was inserted near the posterior coxal cavities, perhaps into the coxal cavities of the middle or hind legs or in the area between them (Fig. 66).
Oviposition was not always fully successful. Of a total of 25 attempts observed, 17 were completed, 4 were initiated but ended with the wasp and the ant -still joined- falling to the ground, and in the other 4 cases the wasp failed to grasp the ant and flew away immediately. Hence, the grasping of the ant appears to be a critical moment of the oviposition process. Sometimes the wasp’s head hit on the ant’s pronotum instead of its mesonotum, or the wasp attacked an ant that was not in a vertical position. In these circumstances it had more difficulty holding the ant, whose vigorous movements usually resulted in oviposition failure. Other times the first hit of the wasp’s head, together with the strong grasping of its fore legs, caused the ant to detach from the surface and fall down with the wasp.
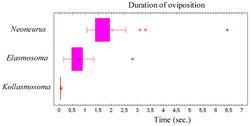
The whole oviposition behaviour of Neoneurus vesculus sp. n. (comprising the grasping of the ant by the wasp and the insertion of the ovipositor, until taking flight) lasted a mean of 2.023 seconds (95% confidence interval: 1.352–2.694; N = 17; SE = 0.317), with a median of 1.507 seconds (interquartile range: 1.377–1.927; Fig. 79). Three outlier observations corresponded to ovipositions lasting more than 3 seconds due to the ant’s vigorous movements which made it difficult for the wasps to bend their metasomata towards the postero-lower part of the ants’ mesosomata.
On one occasion a strange behaviour was observed. One Neoneurus hovering over the nest entrance alighted on the tree trunk, turning and resting, 2 cm away from a worker. The ant approached and touched the apex of the wasp’s metasoma with its antenna. Then the wasp curved its metasoma inward extruding the ovipositor. Finally, the ant attacked the wasp, held it by the wings, and transported it into the nest.
Discussion
The described oviposition behaviour of Neoneurus vesculus sp. n. fits well with the raptorial function predicted by Shaw for the modified morphology of the genus Neoneurus. The head spinules may fix the position of the wasp when its head hits the ant’s mesonotum and the robust tibia are suitable for grasping the ant’s mesosoma by the mesopleura. The location of the wasp when alighting on the ant, and the final arrangement of its body, allow the insertion of the ovipositor into the postero-lower part of the ant’s mesosoma. These facts call for a re-examination, with high speed photography or video, of the oviposition behaviour of Neoneurus mantis in order to confirm the alighting and oviposition of this species in the ant metasoma, as mentioned by Shaw (1993)[2]. It may be remarked that this author several times dissected the ant’s metasoma following the wasp’s oviposition, and could not find the wasp’s eggs. The possibility is open that Neoneurus mantis, and other species of the genus, have a similar oviposition behaviour to that of Neoneurus vesculus sp. n., and hence that the eggs are laid in the ant’s mesosoma.
Diagnosis
Fore tibia of female about 4.0 times as long as wide, distinctly narrowed basally, with long carina and below it a double row of small pegs and with a wide triangular anterior subbasal tooth; mesosoma extensively marked with pale yellowish patches; metasoma brownish-yellow, with first tergite entirely blackish and most tergites basally and apically dark brown, fore femur curved in dorsal view; fore spur nearly straight and robust; facial tubercles small and facial bristles 0.2 times as long as pedicellus, distance between bristles about 1.2 times width of scapus. Runs in the key by Shaw (1992)[1] to Neoneurus pallidus Shaw, 1992, from Canada (Ontario) and USA (Maryland, Michigan, North Carolina, Virginia and Colorado), but the new species has the third and fourth antennal segments pale yellowish (dark brown (except base of third segment) in Neoneurus pallidus); first metasomal tergite entirely blackish (dark yellowish-brown and medially irregularly black), apex and base of second-sixth tergites dark brown (entirely pale yellowish-brown except dark yellowish-brown base of second tergite), fore tibia with row of small pegs below carina (below carina largely smooth, but a few small pegs near apex), fore tibia of female 4.0 times longer than its maximum width in lateral view (4.5 times) and fore tibia of female distinctly narrowed basally (slightly narrowed; Fig. 4 in Shaw 1992[1]).
Description
Holotype, ♀, length of body 2.8 mm, of fore wing 1.8 mm.
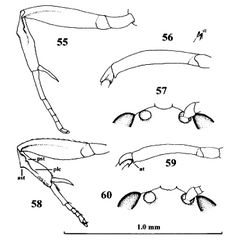
Head. Length of third segment of antenna 1.1 times fourth segment, length of third, fourth and penultimate segments 5.3, 4.8 and 2.5 times their width, respectively and basal segments without distinct setae; facial tubercles small and facial bristles 0.2 times as long as pedicellus, distance between bristles about 1.2 times width of scapus (Fig. 60); length of eye 1.5 times temple in dorsal view; vertex superficially granulate, with few superficial punctures and a satin sheen; temples directly narrowed behind eyes; OOL:diameter of ocellus:POL = 6:3:7; length of malar space 0.13 times height of eye.
Mesosoma. Length of mesosoma 1.4 times its height; mesoscutum superficially punctulate-granulate, but medio-posteriorly densely granulate; precoxal sulcus only medially impressed and with a few rugae; mesopleuron superficially granulate, but postero-dorsally shiny and largely smooth; mesosternal sulcus finely crenulate, narrow and rather shallow; metanotum with a median carina, not protruding dorsally; propodeum finely granulate and with some rugulae, dorsal face longer than posterior face, with satin sheen, only dorsally with a median carina and no medial areola, flat antero-medially and its spiracle small and far in front of middle of propodeum.
Wings. Fore wing: parastigma medium-sized (Fig. 61); basal half of wing nearly as densely setose as its distal half. Hind wing: wing membrane moderately setose basally.
Legs. Hind coxa nearly smooth, dorsally partly superficially micro-granulate; fore coxa flat ventrally; all tarsal claws slender and simple; length of femur, tibia and basitarsus of hind leg 3.9, 9.2 and 5.3 times their width, respectively; fore femur curved in dorsal view, compressed and apically with small tooth; anterior subbasal tubercle of fore tibia wide triangular (Fig. 58) and longitudinal carina of tibia at basal 0.6, bearing a small posterior subbasal tooth and apical half curved, followed by a row of small slender pegs, area of tibia in between subbasal teeth concave (Fig. 59); fore tibia 4.0 times longer than its maximum width in lateral view; fore tibial spur nearly straight and 0.9 times as long as fore basitarsus and 0.5 times fore tibia (Fig. 58); spurs of hind tibia acute apically, their length 0.7 and 0.6 times hind basitarsus.
Metasoma. Length of first tergite 1.4 times its apical width, its surface with satin sheen, granulate with some rugulae posteriorly, basally flat, medially convex and its spiracles slightly protruding and near middle of tergite; second tergite superficially granulate and anteriorly with some oblique rugulae; second metasomal suture obsolescent; remainder of metasoma largely smooth and compressed; setae of metasoma spread, short, but tergites glabrous anteriorly; second tergite with sharp lateral crease; length of ovipositor sheath 0.05 times fore wing.
Colour. Dark brown or blackish; face, clypeus, labrum, malar space, temple ventrally, pronotal side postero-dorsally and ventrally, frons antero-laterally, propleuron, palpi, coxae, trochanters and trochantelli white or ivory; four basal segments of antenna, remainder of legs (but hind tibia and tarsus brown and telotarsi dark brown), tegulae, mesoscutum antero-laterally and a W-shaped patch posteriorly, scutellum (except dark medial patch), mesopleuron antero-dorsally and medially, mesosternum posteriorly, second-fifth metasomal tergites (but anteriorly and posteriorly dark brown) and sixth-eighth tergites pale yellowish; veins pale brown; parastigma, pterostigma and 1-R1 largely dark brown; wing membrane slightly infuscate.
Variation. Length of body 2.6–3.0 mm, of fore wing 1.8–1.9 mm, all females have 16 antennal segments; mesoscutum medially, mesopleuron antero-medially and scutellum may be dark brown; third and fourth antennal segments pale yellow or brownish.
Etymology
From “vesculus” (Latin for “weak, little, poor”) because this new species has poorly developed facial bristles.
Original Description
- Gómez Durán, J; van Achterberg, C; 2011: Oviposition behaviour of four ant parasitoids (Hymenoptera, Braconidae, Euphorinae, Neoneurini and Ichneumonidae, Hybrizontinae), with the description of three new European species ZooKeys, 125: 59-106. doi
Other References
- ↑ 1.0 1.1 1.2 Shaw S (1992) Seven new North American species of Neoneurus (Hymenoptera: Braconidae). Proceedings of the Entomological Society of Washington 94 (1): 26-47.
- ↑ 2.0 2.1 2.2 Shaw S (1993) Observations on the ovipositional behaviour of Neoneurus mantis, an ant-associated parasitoid from Wyoming (Hymenoptera: Braconidae). Journal of Insect Behaviour 6 (5): 649-658. doi: 10.1007/BF01048130
Images
|