Leucochrysa (Nodita) camposi
| Notice: | This page is derived from the original publication listed below, whose author(s) should always be credited. Further contributors may edit and improve the content of this page and, consequently, need to be credited as well (see page history). Any assessment of factual correctness requires a careful review of the original article as well as of subsequent contributions.
If you are uncertain whether your planned contribution is correct or not, we suggest that you use the associated discussion page instead of editing the page directly. This page should be cited as follows (rationale):
Citation formats to copy and paste
BibTeX: @article{Catherine2011ZooKeys92, RIS/ Endnote: TY - JOUR Wikipedia/ Citizendium: <ref name="Catherine2011ZooKeys92">{{Citation See also the citation download page at the journal. |
Ordo: Neuroptera
Familia: Chrysopidae
Genus: Leucochrysa
Name
Leucochrysa camposi (Navás, 1933) – Wikispecies link – ZooBank link – Pensoft Profile
- Nodita camposi Navás, 1933: 197–8, Fig. 17 (Lectotype: MNHN, male, examined; original description: “Ecuador: Guayaquil, Julio de 1932. Campos R. Leg.”). Navás (1934b: 16)[1] [collection record: “Guayaquil (Ecuador), 1933. Campos leg.”; specimen in MNHN]; Penny (1977: 25)[2] [species list].
- Leucochrysa (Nodita) camposi (Navás). First combination in Leucochrysa (Nodita) apparently by Brooks and Barnard (1990: 277)[3] [species list]. Freitas and Penny (2001: 287)[4] [misidentified; see Leucochrysa (Nodita) azevedoi, above]; Oswald (2007)[5] [catalog listing, nomenclature]; Legrand et al. (2008: 121)[6] [lectotype designation, information on type].
- Nodita morenoi Navás 1934a[7]: 157–158 (Lectotype: MNHN, male, examined; original description: “Équateur, Quito, R. Benoist, 1930. Mus. París.”). Synonymized by Legrand et al. (2008: 154)[6]. Here: Synonymy reversed (see below).
Type material
The Nodita camposi lectotype, a male, is badly discolored, but its wings are in good condition, its body is mature, and the genitalia are well sclerotized. A female specimen collected one year later, at the same locality as the type, is also mature.
Diagnosis
Leucochrysa (Nodita) camposi can be distinguished from Leucochrysa (Nodita) azevedoi by its somewhat larger size and more robust appearance (see above) and genitalic characters (male and female) (Figs 8–10). In the male Leucochrysa (Nodita) camposi, the gonarcal arms are narrowly arched; the gonarcal arms extend perpendicularly from the gonarcal bridge, not laterally as in Leucochrysa (Nodita) azevedoi. The gonocornua are located distally on the gonarcal bridge, well outside the span of the mediuncus, and they extend laterally away from the gonarcal bridge. Like Leucochrysa (Nodita) azevedoi, the mediuncus is heavy apically and its beak is borne on a plate (arcessus) that bends back toward the gonarcal arch; however, the membrane extending from the beak does not have distinct arms or patches of setae, as are found on Leucochrysa (Nodita) azevedoi. The hypandrium internum is delicate and has a very narrow U-shape. The Leucochrysa (Nodita) camposi female can be recognized by its bulbous spermatheca, asymmetrical bursa that extends from its left side over the spermatheca and part of the bursal duct, and its highly coiled bursal duct that extends well into the sixth abdominal segment. The bursal glands are elongate, branching and ribbon-like.
Adult description
Head: 1.9 mm wide (including eyes); ratio of head width to eye width = 2.1:1. Vertex raised, with smooth surface, four prominent, circular muscle attachment scars along posterior margin, without setae. Antenna 33.3 mm (~1.5 times length of forewing); scape longer than broad, (0.47–0.50 mm long, 0.36–0.39 mm wide), width = 4.2–4.5× distance between scapes, with ~four long setae distally on dorsal surface, shorter setae laterally; lateral margin fairly straight, mesal margin straight basally, curved outward distally; pedicel ~0.17 mm long, ~0.17 mm wide (at widest point); proximal flagellomeres short (segments 1, 2, 3: length = 1.2–1.5 times width), with three concentric rings of setae; middle and distal segments becoming longer (segments 8–10: length = 1.5–1.8 times width; distal segments: length = 2.1 times width), with four concentric rings of setae. Distance between scapes 0.09 mm; distance between tentorial pits 0.65 mm; length of frons (midway between scapes – midway between tentorial pits) 0.50 mm. Frons relatively flat mesally, with scalloped fold below toruli; surface smooth. Clypeal margins straight; surface slightly textured, not horizontally striated. Labrum with distal margin slightly indented mesally; dorsal surface smooth, setose distally; sides rounded. Ratio of genal length to distance between tentorial pits = 0.31:1.
Head coloration. Specimen largely discolored with age. Antennae: scape amber colored mesally, noticeably darker laterally, probably a broad reddish brown dorsolateral stripe extending full length of scape; pedicel probably with brownish band distolaterally; flagellum cream with golden brown setae, basal ~20 antennomeres with large dark brown to black mark ventrally, fading on antennomeres ~20–28; marks forming prominent dark stripe ventrally. Vertex with raised central area discolored, with thin reddish brown streak across frontal margin, trace of large triangular red mark laterally; area between raised area and eyes maybe marked with red. Frons, clypeus white, unmarked; labrum cream to amber; gena discolored, possibly red throughout. Maxilla, maxillary palpi, labium, labial palpi white to cream.
Thorax: Cervix small, largely withdrawn below prothorax, discolored, with small reddish lateral marks. Prothorax (sclerotized region) 0.95 mm long; 1.3 mm wide; ratio of length : width = 0.74:1; prothorax (extended) 1.3 mm long; setae thin, long, golden; discolored, with pair of anterolateral spots, pair of elongate, sublateral, red marks with irregular margins. Mesothorax, metathorax discolored; mesoprescutum with pair of red marks mesally; mesoscutum with pair of red marks on anteromesal flank; metascutum with pair of thin red submesal streaks from middle to posterior margin. Legs unmarked, with golden setae, described by Navás as: greenish, tarsi probably amber.
Wings: Forewing 21.3–21.6 mm long, 8.1–8.2 mm wide (at widest point); ratio of length : maximum width = 2.7:1. Costal area slightly expanded basally; tallest costal cell (#8, 9) 1.1 mm tall, 2.1 times width, 0.2 times width of wing (midwing). First intramedian cell triangular, 0.7 times width of third median cell. First radial crossvein distal to origin of radial sector (Rs); radial area (between Radius and Rs) with single row of 17 closed cells; tallest cell (#6) 3.0 times taller than wide. No crassate veins; 5 b cells. Two series of gradate veins; 10–11 inner gradates, 10–11 outer gradates; 6–8 b’ cells. Three intracubital cells (two closed). Membrane clear, except posteroapical faint tinge of brown streaked across bases of forked distal veinlets; stigma golden, with small brown mark basally. Veins green, with dark brown to black on tips of costal veinlets, basal segment of Rs (to first posterior crossvein), two basal radial crossveins, midsection of Rs and radial crossveins above, bases of marginal forks.
Hindwing 18.5–18.8 mm long, 5.5–6.1 mm wide. Two series of gradate veins; 8 inner, 9 outer; 15 radial cells (counted from origin of Radius, not false origin). Four to five b cells (including small b1 cell); five b’ cells beyond second intramedian cell; two intracubital cells (one closed). Stigma golden, marked with brown distally; membrane clear with posteroapical margin streaked with faintly brown tinge. Veins green, except midsection of Rs, tips of marginal forks (posteroapical margin of wing) dark brown to black.
Abdomen (male type only; female abdomen damaged): Distal segments (beyond A4) expanded; pleural region (P6) ca. 2.75 times height of sternite (S6). Setae on tergites, sternites, moderately long, slender; setae on pleura short, small; microsetae moderately dense, small throughout. S6 approx. 1.3 times longer than tall, S7 approx. 1.1 times longer than tall (lateral view). Tergites cap-like, with lighter setae and longer microsetae than on sternites. Spiracles oval externally; atria not enlarged. Coloration: specimen discolored.
Male [gonarcal complex separated from abdomen, connecting membrane remains attached to gonarcus]: Callus cerci ~round, 0.21–0.25 mm diameter (range), with 39 long, thin, trichobothria. Sternites 3–8 (not S1 or S2) with microtholi. T9+ectoproct short, truncate distally, fused mesally, midline without deep cleft, setae long, moderately slender throughout; ventral section of T9+ectoproct with elongate proximal extension reaching full length of A8; proximal section well sclerotized, with apodeme heavy, extending around proximal margin of callus cerci. S8+9 fused, without suture; S9 without microtholi; S8 much shorter, slightly taller than S9; S8+9 (lateral view) with proximal, distal margins straight, with acute ventral apices, rounded dorsal apices, slightly <1/2 height of proximal margin. Setae on S9 very slightly heavier than those on S5-S8; terminus of S9 without gonocristae. Subanal region membranous, with small striations around anus, no setae. Gonarcal complex attached to gonarcal bridge by clear, smooth membrane around base of gonocornua; gonocornua protruding through membrane; section of membrane below mediuncus confluent with gonosaccus below mediuncus, holding hypandrium internum distally. Gonarcus robust, narrowly arcuate, with lateral apodemes extending perpendicularly from gonarcal bridge; gonarcal apodemes robust, stiff, lateral surface slightly concave, expanded distally (lateral view). Gonocornua broadly attached to gonarcal bridge distal to mediuncus, broad basally, tapering distally, with straight margins, rounded tips. Mediuncus largely sclerotized; base with membranous attachment to dorsum of gonarcal bridge, mesal to gonocornua; basal section with pair of shallow, elongate depressions, acute ridge between, becoming flattened, expanded distally; terminus rounded laterally, scalloped frontally; ventral surface (below expanded tip of mediuncus) with elongate, recurved, mesal beak; sides of mediuncus membranous, connecting below mediuncus, forming internal pouch below beak, above gonosaccus. Gonosaccus clear, smooth, folded membrane, bearing ~3 pairs of robust, medium-length gonosetae on small chalazae. Entoprocessus, tignum, gonapsis, pseudopenis, spinellae, gonocristae absent. Hypandrium internum with narrow U-shape, apex rounded, delicate, lightly sclerotized; comes very faint.
Female: Callus cerci round, 0.26 mm maximum diameter, with 39 slender trichobothria. Tergite 8 roughly quadrate (lateral view), similar in depth to T6. Tergite 9+ectoproct elongate; posterior margin: dorsal one-third straight, perpendicular to dorsal margin of tergite, then indented, angled inward, straight; ventral margin rounded, extending to depth of gonapophyses laterales. Sternite 7 largely damaged. Gonapophysis lateralis angled dorsally, rounded distally, ventrally, ~0.60 length of T9+ect; inner membranous surface not expandable, with sparse, small setae on inner surface. Colleterial gland missing but short, thick duct attached immediately before transverse sclerification. Bursa copulatrix concave, shield-like, lightly folded, thick membranous sac, extending anteriorly over spermatheca, into mid-section of S7, also extending, asymetrically on left side of S7 above ca. basal one-half of coiled bursal duct; with pair of slender, elongate, branching, ribbon-like bursal glands, one connected on either side of bursa, near interior tip of bursa. Bursal duct flat to thickened, coiled, robust, tapering anteriorly to A6, then coiling back on itself to bursa; membranous throughout. Spermatheca broad, thick, U-shaped tube, thick walled with wide circular opening facing subgenitale, enlarged tire-shaped bulge at open end, prominent slit along entire dorsum, opening to bursal duct (0.16 mm diameter at mouth, narrower at midsection), ~0.5 mm long, with narrow (0.04 mm), moderate length (0.12 mm) invagination. Spermathecal duct uncolored, originating on dorsal surface of spermatheca at terminus of dorsal slit, making two sharp bends, a U-shaped bend in subgenitale, then glandulose thicked portion making broad bend back toward spermatheca. Subgenitale large, rounded, with rigid, folded, heavy membraneous texture, terminus broad, flat-surfaced, bilobed dorsally (heart shaped in posterior view), with large, flat transverse depression mesally below lobes, with ventrally protruding tip; small ventral fold above S7 without setae.
Larvae and biology
Unknown.
Distribution
Currently known only from Ecuador (Guayaquil, Quito).
Adult specimens examined
In addition to the type listed above, we examined two female specimens from the type locality; both are in the MNHN. One was collected in 1933 and was reported by Navás (1934b: 16)[1]. The other specimen bears a determination label reading “Nodita Azevedoi Nav, P. Navás S.J. det.”; it was reported by Navás (1928: 111)[8]. The descriptions above are based on the male type and the female specimen reported by Navás in 1934.
Etymology
Navás named the species in honor of its collector, D. Francisco Campos R. of Guayaquil, Ecuador. Campos provided Navás with numerous lacewing specimens from Ecuador.
Taxon Treatment
- Catherine A., T; Gilberto S., A; Maurice J., T; 2011: Nomenclatorial changes and redescriptions of three of Navás’ Leucochrysa (Nodita) species (Neuroptera, Chrysopidae) ZooKeys, 92: 9-33. doi
Other References
- ↑ 1.0 1.1 Navás L (1934b) Insectos suramericanos. Octava [VIII] serie. Revista de la Real Academia de Ciencias Exactas Fisicas y Naturales de Madrid 31:9-28.
- ↑ Penny N (1977) Lista de Megaloptera, Neuroptera e Raphidioptera do México, América Central, ilhas Caraíbas e América do Sul. Acta Amazonica 7(4) (Suplemento): 1–61.
- ↑ Brooks S, Barnard, P (1990) The green lacewings of the world: a generic review (Neuroptera: Chrysopidae). Bulletin of the British Museum of Natural History, Entomology 59:117-286.
- ↑ Freitas S, Penny, N (2001) The green lacewings (Neuroptera: Chrysopidae) of Brazilian agro-ecosystems. Proceedings of the California Academy of Sciences 52:245-395.
- ↑ Oswald J (2007) Neuropterida Species of the World. Version 2.0. http://lacewing.tamu.edu/Species-Catalogue/. [accessed on Nov. 17, 2010]
- ↑ 6.0 6.1 Legrand J, Tauber C, Albuquerque G, Tauber M (2008) Navás’ type and non-type specimens of Chrysopidae in the MNHN, Paris [Neuroptera]. Revue française d’Entomologie 30:103-183.
- ↑ Navás L (1934a) Insectos suramericanos. Novena [IX] serie. Revista de la Real Academia de Ciencias Exactas Fisicas y Naturales de Madrid 31:155-184.
- ↑ Navás L (1928). Insectos neotropicos. 4.a serie. Revista Chilena de Historia Natural 32:106-128.
Images
|


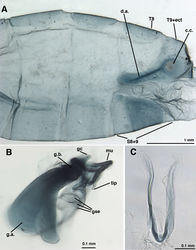
![Figure 9. Leucochrysa (Nodita) camposi, Male gonarcus, series of views. The arrows on the lateral view, A, indicate the direction of the views in B–D. (Guayaquil, Ecuador, Lectotype, MNHN). A Lateral view B Gonarcus, view from beneath, with gonocornua extending upward above gonarcal bridge [Note heavy membrane around base of gonocornua] C Gonarcus, view from beneath, with gonocornua extending upward above gonarcal bridge, focus on frontal surface of mediuncus and gonoseta on membrane beneath gonarcus D Gonarcus, view from above, with gonocornua extending upward above gonarcal bridge, focus on junction of mediuncus with gonarcal bridge. Abbreviations: gc gonocornu gse gonoseta g.b. gonarcal bridge mu mediuncus tip tip of mediuncus beak.](/w/media/thumb/7/7c/ZooKeys-092-009-g009.jpg/250px-ZooKeys-092-009-g009.jpg)
![Figure 10. Leucochrysa (Nodita) camposi, Female [MNHN, Nontype Navás specimen]. A Terminalia, dissected, lateral view from right B Bursal duct, part of bursa copulatrix, lateral view from left C Subgenitale, posterior view D Spermatheca beneath bursa copulatrix, ventral view E Spermatheca beneath bursa copulatrix, ventrolateral view F Spermatheca, dorsal view, through bursal copulatrix [Note dorsal slit.]. Abbreviations: b.c. bursa copulatrix b.d. bursal duct b.gl. bursal gland d.o. dorsal slit or opening from spermatheca to bursa copulatrix inv spermathecal invagination sg subgenitale sp spermatheca.](/w/media/thumb/c/c4/ZooKeys-092-009-g010.jpg/250px-ZooKeys-092-009-g010.jpg)