Lepidasthenia loboi
| Notice: | This page is derived from the original publication listed below, whose author(s) should always be credited. Further contributors may edit and improve the content of this page and, consequently, need to be credited as well (see page history). Any assessment of factual correctness requires a careful review of the original article as well as of subsequent contributions.
If you are uncertain whether your planned contribution is correct or not, we suggest that you use the associated discussion page instead of editing the page directly. This page should be cited as follows (rationale):
Citation formats to copy and paste
BibTeX: @article{Salazar-Vallejo2015ZooKeys, RIS/ Endnote: TY - JOUR Wikipedia/ Citizendium: <ref name="Salazar-Vallejo2015ZooKeys">{{Citation See also the citation download page at the journal. |
Ordo: Phyllodocida
Familia: Polynoidae
Genus: Lepidasthenia
Name
Lepidasthenia loboi Salazar-Vallejo & González & Salazar-Silva, 2015 sp. n. – Wikispecies link – ZooBank link – Pensoft Profile
- Lepidasthenia esbelta: Barnich et al. 2012[1]: 406–407 (non Amaral & Nonato, 1982).
Type material
Southwestern Atlantic, Argentina. Cerro Avanzado, 16 km southward from Puerto Madryn (42°49'S, 65°04'W), Golfo Nuevo. Holotype (ECOSUR 176), and 12 paratypes, rocky shore, intertidal, in mudstone, within tubes of Thelepus antarcticus Kinberg, 1867, coll. 27 Feb. 2013, J.M. Orensanz, N.E. González & S.I. Salazar-Vallejo [Paratypes: Two (ECOSUR 177), 45–64 mm long, 5–6 mm wide, 83–97 chaetigers; two paratypes (LACM 7040), 30–32 mm long, 4 mm wide, 63–77 chaetigers; two paratypes (MACN), 40–58 mm long, 4.0–4.5 mm wide, 78–90 chaetigers; three paratypes (MZUSP 2857), 12–42 mm long, 2–4 mm wide, 37–83 chaetigers; two paratypes (ZUEC 17781, 17782), 36–39 mm long, 4 mm wide, 79–80 chaetigers.
Additional material
Southwestern Atlantic, Argentina. One specimen (ECOSUR), San Antonio Oeste (40°44'S, 64°57'W), Golfo San Matías, 3 m, coll. 10 Oct. 2005, J.M. Orensanz (30 mm long, 4 mm wide, 70 chaetigers). Six anterior fragments (ECOSUR), Cerro Avanzado, 16 km southward from Puerto Madryn (42°49'S, 65°04'W), rocky shore, intertidal, in mudstone, with Thelepus antarcticus Kinberg, 1867, coll. 27 Feb. 2013, J.M. Orensanz, N.E. González & S.I. Salazar-Vallejo.
Description
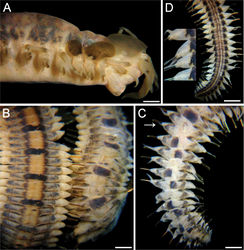
Holotype (ECOSUR 176) twisted, almost complete (without anal cirri). Body 64 mm long, 5 mm wide (at chaetiger 1, without chaetae), 99 chaetigers. Antennae, palps and tentacular cirri pale. Dorsal cirri with blackish cirrophore, cirrostyles with subdistal blackish ring, tips pale (Fig. 1A). Dorsum with almost continuous thick, lateral, longitudinal dark-brown bands; bands continuous in chaetigers 1–4, medial areas paler; chaetiger 5 pale, alternating with blackish transverse band (Fig. 1B, C). First transverse band (chaetiger 6) slightly longer than corresponding segment, followed by irregular transverse bands occupying slightly more than half of segment length along 6 chaetigers, middorsal areas with irregular brownish spots, bands then alternating to chaetiger 21, thereafter darker bands every three segments but intermediate segments paler, maculated. First elytra greyish, largest (Fig. 1A); following ones blackish, markedly smaller. Venter smooth; anterior third pale, posterior two-thirds with discontinuous darker, blackish bands along nephridial lobes areas; midventral region slightly less pigmented, ventral chord area paler (Fig. 1D). Far posterior segments with darker pigmentation ventrally. Nephridial papillae projecting, dark. Prostomium with eyes black, medium-sized (as wide as antennal width), central on prostomium; anterior eyes more separated than posterior ones (Fig. 2A). Medial antenna slightly longer than laterals; ceratophores of similar width, slightly longer than wide; ceratostyles tapered, with long tips. Palps 3–4 times thicker than antennae, right one 2.5 times longer than medial antenna, left one regenerating. Segment 1 with tentacular cirri 1.3 times as long as, and slightly thicker than, antennae, tapered, with long tips mucro. Elytra on segments 2, 4, 5, alternating with dorsal cirri to chaetiger 26, thereafter on every three segments but last 7 segments more irregular. First pair of elytra largest, covering prostomium and middorsal region, grayish, slightly darker around junction area, laterally with a paler, thin area. Second pair of elytra blackish, oval, less than half as large as first elytra, slightly overlapping anterior elytra, not covering middorsal region, laterally with a paler, thin area. Third pair of elytra blackish, subcircular, less than half as large as second elytra, non-overlapping with previous elytra, not covering middorsal region. Following elytra with same pigmentation, progressively reducing in size, up to chaetiger 20, about twice as large as junction area.
Parapodia sub-biramous throughout body. Notopodia reduced to a projecting, digitate lobe, reducing in size posteriorly. Neuropodia projecting lobes throughout body, neurochaetal lobes truncate or rounded. Dorsal cirri with cirrophores blackish, about as long as wide, cirrostyles tapered, with long tips, longer anteriorly, slightly reducing in length and pigmentation posteriorly, about twice as long as neuropodium. Ventral cirri small, tapered, basal-half blackish, tips mucronate, arising at base of parapodia, about as long as half neuropodial length.
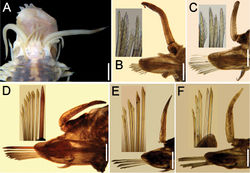
Notopodia without notochaetae. Neurochaetae of different size and shape. Anterior chaetigers with about 15 neurochaetae per bundle, of similar width, smaller ventrally, each with bidentate tips, accessory tooth smaller, directed distally, and 10 or more series of subdistal lamellae (Fig. 2B, C). Medial chaetigers with one giant (thicker, more than twice as wide as other ones), brownish, superior neurochaeta with series of 5–6 tiny spines, tips unidentate, and about 10 thinner bidentate chaetae with series of 5–6 lamellae, becoming thinner and smaller ventrally (Fig. 2D, E). Posterior neuropodia with 1–2 slightly wider, superior chaetae and 4–5 thinner neurochaetae (Fig. 2F). Neuraciculae hyaline, tapered.
Posterior region tapered; pygidium truncate, anus dorsal. Nephridial papillae from chaetiger 9; anterior region with papillae pale, smaller along anterior body half, progressively larger and darker in posterior body half.
Pharynx (observed in some paratypes) with marginal prismatic papillae, upper ones larger, 9 upper and 9 lower. Two pairs of dark brown jaws.
Etymology
This species name honors the late José María Orensanz, in recognition of his many contributions to the study of Southwestern Atlantic and Antarctic polychaetes, of his continued support of our research dreams, and for his participation in the field trip that collected the species. The specific epithet is derived from his nickname, Lobo, and is a noun in apposition.
Type locality
Cerro Avanzado rocky shore, intertidal, Puerto Madryn (42°49'S, 65°04'W), Golfo Nuevo, Argentina.
Variation
Paratypes 12–64 mm long, 2–6 mm wide, 37–99 chaetigers. Smallest specimen with transverse bands restricted to anterior region; larger specimens more heavily pigmented and showing variation in the amount of spots or darkening of paler areas between successive transverse bands. Intensity of pigmentation increased in larger specimens, and in some (including holotype), posterior region had an irregular pattern probably due to imperfect regeneration, which is rather uncommon in other errant polychaetes (Yáñez-Rivera and Méndez 2014[2]).
Remarks
Lepidasthenia loboi sp. n. has been confused with Lepidasthenia esbelta Amaral & Nonato, 1982, described from southern Brazil because both live with Thelepus, have similar size and pigmentation patterns, and giant neurochaetae. However, they differ in several diagnostic features such as the size of eyes, the type of cephalic and parapodial appendages, size of anterior elytra, topology of parapodial cirri, and tips of neuraciculae. In Lepidasthenia loboi sp. n. eyes are of the same size, cephalic and parapodial cirri have long tapered tips, second pair of elytra is larger than third, ventral cirri arise basally to neuropodia such that they do not reach the tips of the chaetal lobe, and neuraciculae are tapered. On the contrary, in Lepidasthenia esbelta posterior eyes are larger than the anterior ones, cephalic and parapodial appendages are subdistally swollen, the second and third pairs of elytra are of the same size, ventral cirri are medially placed such that their tips reach the tips of the neurochaetal lobe, and the neuroaciculae have falcate tips.
Another species has been recorded from Brazil by Nonato and Luna (1970)[3], and by Amaral and Nonato (1982)[4]: Lepidasthenia virens (Blanchard in Gay, 1849). These records indicate a lepidastheniin without notochaetae that resembles Lepidasthenia loboi because of the type of antennae and tentacular cirri, although palps are shorter than antennae, and by the relative size and position of parapodial cirri, although they illustrated a mature female with hypertrophied dorsal cirrophores. They gave no further detail and the affinities between these two species cannot be clarified. However, two issues deserve comments.
First, Lepidasthenia virens was described briefly with material from Calbuco (41°46'S, 73°08'W), Chiloé, Chile. The description and illustration indicates that elytra are large enough to touch each other along the body but while leaving the middorsal surface uncovered (Blanchard 1849[5]:16, Pl. 1, Fig. 2: “… dejando descubierta la porción del medio del dorso, y en cuanto á la longitud del cuerpo apenas si se cubren”). Ehlers (1901[6]:54, Pl. 3, Figs 10–16) described Lepidasthenia irregularis with material from the same locality; this species has elytra touching each other, leaving the middorsal surface bare, and notochaetae are present in anterior parapodia. If Lepidasthenia virens and Lepidasthenia irregularis are synonyms then they both belong in Lepidametria by having notochaetae and large elytra overlapping or touching successive ones.
Second, Hartman (1939[7]: 46) noticed this synonymy but because she studied material from a more tropical region, her illustrations do not match Ehlers’ ones. Her specimens have no notochaetae, and neurochaetae are very abundant (ca. 20 per bundle vs about 10 per bundle). Consequently, the Eastern tropical Pacific material belongs to another, probably undescribed species, and they differ from true Lepidasthenia virens (? = Lepidasthenia irregularis).
Ecological notes
Thelepus antarcticus Kinberg, 1867 builds its tubes with a mucoid protein forming a semi-transparent matrix with attached fragments of shells or other calcareous fragments. Tubes run inside rock crevices or fractures and are difficult to track individually because they break when the rock is fragmented. There were 34 Thelepus antarcticus specimens plus six belonging to two other terebellid species, making it the most frequent thelepodid (or terebellid) in the rocky intertidal environment. About half of Lepidasthenia loboi specimens remained inside Thelepus tubes, whereas the others left the tubes as the rock was broken. In total, there were 19 Lepidasthenia loboi specimens taken at Cerro Avanzado, and there were polynoids in only one-third of the Thelepus tubes, half the rate of association between Thelepus crispus and Halosydna brevisetosa found by Morgan (1974)[8]. It would be interesting to conduct a more detailed study to find out what are the functional relationships between Thelepus antarcticus and Lepidasthenia loboi sp. n. Some specimens exhibited regeneration of palps, antennae, or both, indicating there must be some interactions with other invertebrates, possibly other scale-worms. Some of the anterior fragments were dissected for gut contents but none were found.
Distribution
The specimens were found in two localities in two southern Argentina Gulfs: Cerro Avanzado, Puerto Madryn, Golfo Nuevo, and San Antonio Oeste, Golfo San Matías, but might co-occur with Thelepus antarcticus throughout its distribution. Kinberg (1867[9]: 345) described Thelepus antarcticus from the intertidal in York Bay, Bucket Island, Magellan Strait. Hartman (1966[10]:109) and Rozbaczylo et al. (2006[11]: 83) regarded it as a junior synonym of Thelepus plagiostoma (Schmarda, 1861: 41), described from New Zealand. However, this synonymy was not based upon a study of type material so these two species must be regarded as distinct until a future comparison involving type specimens indicates otherwise. The distribution of Thelepus antarcticus would correspond to Patagonian shores, along southern Chile and Argentina, in intertidal to shallow water bottoms.
Key to species of Lepidasthenia Malmgren, 1867 with giant neurochaetae
* A junior synonym of Lepidasthenia virens (Blanchard in Gay, 1849) fide Hartman (1956[12]: 271); they are probably different. Lepidasthenia virens was described from Chiloé, southern Chile, whereas Lepidasthenia ornata is from western Mexico. If Lepidasthenia virens is the same as Lepidasthenia irregularis Ehlers, 1901, both described from the same locality in Chile, and having large notopodia, the latter provided with notochaetae, then both belong in Lepidametria.Original Description
- Salazar-Vallejo, S; González, N; Salazar-Silva, P; 2015: Lepidasthenia loboi sp. n. from Puerto Madryn, Argentina (Polychaeta, Polynoidae) ZooKeys, (546): 21-37. doi
Images
|
Other References
- ↑ Barnich R, Orensanz J, Fiege D (2012) Remarks on some scale worms (Polychaeta, Polynoidae) from the Southwest Atlantic with notes on the genus Eucranta Malmgren, 1866, and description of a new Harmothoe species. Marine Biodiversity 42: 395–410. doi: 10.1007/s12526-012-0117-4
- ↑ Yáñez-Rivera B, Méndez N (2014) Regeneration in the stinging fireworm Eurythoe (Annelida): Lipid and triglyceride evaluation. Journal of Experimental Marine Biology and Ecology 459: 137–143. doi: 10.1016/j.jembe.2014.05.023
- ↑ Nonato E, Luna J (1970) Sobre algunos poliquetas de escama do Nordeste do Brasil. Boletim do Instituto Oceanografico, São Paulo 18: 63–91. doi: 10.1590/S0373-55241969000100008
- ↑ Amaral A, Nonato E (1982) Anelídeos poliquetos da costa brasileira, 3. Aphroditidae e Polynoidae. Conselho Nacional de Desenvolvimento Cientifico e Tecnologico, Brasilia, 46 pp.
- ↑ Blanchard E (1849) Anulares. In: Gay C (Ed.) Historia Física y Política de Chile. Zoología, Tomo 3, Paris, 5–39. http://www.memoriachilena.cl/archivos2/pdfs/mc0019535.pdf
- ↑ Ehlers E (1901) Die Polychaeten des magellanischen und chilenischen Strandes: Ein faunisticher Versuch. Fetschrift zur Feier des Hundertfünfzigjärigen Bestehens der Königlichen Gesellschaft der Wissenschaften zu Göttingen. Weidmannsche Buchhandlung, Berlin, 232 pp.
- ↑ Hartman O (1939) Polychaetous annelids, 1. Aphroditidae to Pisionidae. Allan Hancock Pacific Expeditions 7: 1–155. http://digitallibrary.usc.edu/cdm/ref/collection/p15799coll82/id/15606
- ↑ Morgan J (1974) Physiological and behavior studies of Thelepus crispus and its commensal Halosydna brevisetosa. Portland State University, M. Sc. Thesis, 66 pp. http://pdxscholar.library.pdx.edu/cgi/viewcontent.cgi?article=3010&context=open_access_etds
- ↑ Kinberg J (1867(1866)) Annulata Nova (Anthostomea, Chloraemea, Chaetopterea, Maldanea, Ammocharidea, Ampharetea, Terebellea, Hermellea, Serpulea, Teletusea, Hirudinacea). Ofversigt af Konglelige Vetenskaps-Akademiens Förhandlingar 23: 337–357.
- ↑ Hartman O (1966) Polychaeta Myzostomidae and Sedentaria of Antarctica. Antarctic Research Series 7: 1–158. doi: 10.1029/ar007
- ↑ Rozbaczylo N, Moreno R, Díaz-Díaz O, Martínez S (2006) Poliquetos bentónicos submareales de fondos blandos de la región de Aysén: Clado Terebellida (Annelida, Polychaeta). Ciencia y Tecnología Marinas 29: 71–90.
- ↑ Hartman O (1956) Polychaetous annelids erected by Treadwell, 1891-1948, together with a brief chronology. Bulletin of the American Museum of Natural History 109: 243–310. http://www.biodiversitylibrary.org/bibliography/89272#/summary