| Notice: |
This page is derived from the original publication listed below, whose author(s) should always be credited. Further contributors may edit and improve the content of this page and, consequently, need to be credited as well (see page history). Any assessment of factual correctness requires a careful review of the original article as well as of subsequent contributions.
If you are uncertain whether your planned contribution is correct or not, we suggest that you use the associated discussion page instead of editing the page directly.
This page should be cited as follows (rationale):
Sereno P (2012) Taxonomy, morphology, masticatory function and phylogeny of heterodontosaurid dinosaurs. ZooKeys 226 : 1–225, doi. Versioned wiki page: 2012-10-03, version 27691, https://species-id.net/w/index.php?title=Heterodontosaurus_tucki&oldid=27691 , contributors (alphabetical order): Pensoft Publishers.
Citation formats to copy and paste
BibTeX:
@article{Sereno2012ZooKeys226,
author = {Sereno, Paul C.},
journal = {ZooKeys},
publisher = {Pensoft Publishers},
title = {Taxonomy, morphology, masticatory function and phylogeny of heterodontosaurid dinosaurs},
year = {2012},
volume = {226},
issue = {},
pages = {1--225},
doi = {10.3897/zookeys.223.2840},
url = {http://www.pensoft.net/journals/zookeys/article/2840/abstract},
note = {Versioned wiki page: 2012-10-03, version 27691, https://species-id.net/w/index.php?title=Heterodontosaurus_tucki&oldid=27691 , contributors (alphabetical order): Pensoft Publishers.}
}
RIS/ Endnote:
Wikipedia/ Citizendium:
See also the citation download page at the journal. |
Taxonavigation
Ordo: Ornithischia
Familia: Heterodontosauridae
Genus: Heterodontosaurus
Name
Heterodontosaurus tucki Crompton and Charig, 1962 – Wikispecies link – Pensoft Profile
- Heterodontosaurus tucki Crompton and Charig, 1962 – Crompton and Charig (1962[1], fig. 1); Halstead Tarlo et al. (1963[2], fig. 1); Galton (1970[3], fig. 4C); Charig and Crompton (1974, Figs 10, 11); Santa Luca et al. (1976[4], Figs 1, 2); Thulborn (1978[5], Figs 1, 2A); Hopson (1980[6], Figs 4, 6); Santa Luca (1980, Figs 1-23, append. 1); Weishampel (1984[7], Figs 3, 12C); Crompton and Attridge (1986[8], Figs 17.9–17.11); Bakker and Galton (1974[9], fig. 1H); Bakker (1986[10], fig. on p. 453); Galton (1986[11], Figs 6R–T, 15.5B); Weishampel and Witmer (1990[12], Figs 23.3, 23.4); Norman and Weishampel (1991[13], fig. 7); Brett-Surman (1997[14], fig. 24.1B); Norman et al. (2004[15], Figs 18.1, 18.2D, 18.7); Langer and Benton (2006[16], fig. 8A); Butler et al. (2008[17], Figs 1-5); Smith (1997[18], fig. 1); Brusatte et al. (2010[19], fig. 3D); Porro et al. (2011[20], Figs 2, 3, 8a-c); Norman et al. (2011[21], Figs 1–36, append. 3–6)
Holotype
SAM-PK-K337, nearly complete, articulated skull.
Referred material
SAM-PK-K1332, articulated skull and skeleton lacking only a few mid and distal caudal vertebrae; SAM-PK-K10487, anterior portion of a juvenile skull including the left orbit with palpebral and anterior portion of the lower jaws with the predentary; SAM-PK-K1328, partial postcranial skeleton including dorsal and caudal vertebrae, forelimbs and hindlimbs; SAM-PK-K1334, left maxilla with six teeth and portions of adjacent bones preserving the antorbital fenestra, the ventral end of the lacrimal, and the anterior end of the jugal; AMNH 24000, posteroventral portion of skull with articulated lower jaws preserving the posterior half of maxillary and dentary tooth rows, parts of the right jugal, quadratojugal and quadrate, posterior half of the right lower jaw, and traces of the anterior cervical vertebrae.
Type locality
On the mountainside behind the trading store in Tyinindini at an altitude of 1890 m, Transkei (Herschel) District, Cape Province, South Africa; S30°32', E27°32' (Crompton and Charig 1962[1]; Kitching and Raath 1984[22]) (Fig. 1B).
Horizon
Upper Elliot Formation and Clarens Formation (Smith 1990[23]; Knoll 2005[24]); Lower Jurassic, Hettangian to Sinemurian, ca. 200-190 Ma (Crompton and Charig 1962[1]; Santa Luca et al. 1976[4]; Gradstein and Ogg 2009[25]).
Revised diagnosis
Heterodontosaurid ornithischian characterized by the following autapomorphies: (1) cheek tooth crowns subrectangular in cross-section; (2) prominent crown margins and primary ridge resulting in mesial and distal paracingular fossae on labial and lingual faces of maxillary and dentary crowns, respectively; (3) asymmetrical enamel on maxillary and dentary crowns (reduced in thickness on lingual and labial sides of maxillary and dentary crowns, respectively); (4) lacrimal with shallow lateral fossa; (5) jugal with extension of antorbital fossa onto the orbital ramus; (6) jugal flange posteroventrally inclined; (7) trapezoidal anterior surangular foramen; (8) axis and C3 neural spine with lateral flange; (9) C5 and C6 neural spines project anterodorsally; (10) C3-C7 with subcylindrical parapophyses; (11) mid dorsal vertebrae (D6-D10) with Y-shaped transverse processes (for di- and parapophyses); (12) scapulocoracoid foramen absent; (13) humeral epicondyles present; (14) lobe-shaped distal expansion of the iliac preacetabular process; (15) ischial peduncle narrow, columnar; (16) ischial shaft with laterally-directed crescentic flange at mid-length; (17) femoral anterior and greater trochanters coossified; (18) tibiofibulotarsus coossification (possibly variable); (19) tarsometatarsus coossification.
The initial diagnosis of Heterodontosaurus tucki by Crompton and Charig (1962[1]: 1075) listed 10 features, none of which now stand as autapomorphies particular to the species. Some of the listed features, such as an edentulous predentary, are plesiomorphies of broad distribution, whereas others (presence of three premaxillary teeth, arched diastema) now characterize other heterodontosaurids. In a review of ornithischian taxa, Steel (1969[26]: 7-8) listed more than a dozen cranial features in a revised diagnosis, but again none are regarded here as autapomorphies given the diversity of heterodontosaurids now known. The reduction of the cingulum and the presence of a jugal flange, for example, are now known in the recently described South American heterodontosaurid Manidens (Pol et al. 2011[27]). In his description of the postcranial skeleton, Santa Luca (1980[28]: 197) presented a revised diagnosis based solely on postcranial features. Of the 20 features listed, 4 are regarded here as autapomorphies that are currently known in other heterodontosaurids (autapomorphies 13, 17-19; humeral epicondyles; femoral anterior and greater trochanters coossified, tibiotarsus and tarsometatarsus coossification).
The recent revision of the diagnosis in Norman et al. (2011[21]: 187) added 19 cranial and dental autapomorphies to a revised list of postcranial features from Santa Luca for a total of 44 characters. The authors marked 30 of these as potential autapomorphies. Many of these features, however, are problematic as autapomorphies. Of the 12 cranial features cited by Norman et al. (2011)[21], 4 are retained in the revised diagnosis above (autapomorphies 2, 5–7). Some of the features cited by these authors but excluded here are known in other heterodontosaurids, such as the everted rim on the maxilla, presence of a jugal horn and flange, T-shaped quadratojugal, the reduction of the cingulum, and the presence of an external mandibular fossa (Thulborn 1974[29]; Pol et al. 2011[27]). The form of the basal tubera and basipterygoid processes also appear to be similar to that in Manidens (Pol et al. 2011[27]). One of the cited features, “two fingerlike rami” of the surangular, is based on a damaged surangular as discussed below. One cranial feature new to the diagnosis above is the shallow subtriangular fossa on the lateral aspect of the lacrimal (autapomorphy 4) (Fig. 59).
Two of the seven dental features cited by Norman et al. (2011)[21] are comparable to autapomorphy 2 in the diagnosis above. Other dental features cited by these authors are difficult to differentiate from the condition in several other heterodontosaurids, such as the distribution of serrations on caniniform teeth and reduction of the cingulum. The presence of extensive tooth wear, another cited feature, is not regarded here as diagnostic, given the presence of broad wear facets in Lycorhinus, Abrictosaurus and Pegomastax gen. n. sp. n.Dental features new to the revised diagnosis (autapomorphies 1, 3) include the subrectangular cross-section of the cheek teeth (Fig. 49C) and asymmetrical enamel on maxillary and dentary crowns. The distribution and thickness of enamel on the crowns of Heterodontosaurus tucki and other heterodontosaurines is poorly established, and so this may eventually characterize other species. Basal heterodontosaurids such as Echinodon and Fruitadens do not appear to have asymmetrical enamel on the crowns of the cheek teeth.
Six of the 10 postcranial features cited by Norman et al. (2011)[21] as autapomorphic are reformulated above (as autapomorphies 13, 16–19). Other postcranial features cited by these authors are difficult to defend as autapomorphies, such as the number of precaudal or sacral vertebrae (see discussion below), the presence of epipophyses in anterior cervical vertebrae (present in Lesothosaurus), and a narrow scapular blade (present in other heterodontosaurids). Postcranial features new to the revised diagnosis (autapomorphies 8-12, 14, 15) include unusual features of the axial skeleton, the absence of the scapulocoracoid foramen, and the shape of processes in the pelvic girdle.
Description
The aim of the following descriptive comments is to correct and extend where needed available accounts of the skull and skeleton of the best-known heterodontosaurid, Heterodontosaurus tucki. Initial accounts of the skull of Heterodontosaurus were based on two specimens, the holotype (SAM-PK-K337; Crompton and Charig 1962[1]) and a better preserved referred specimen (SAM-PK-K1332; Charig and Crompton 1974[30]; Weishampel 1984[7]). Since that time, additional specimens have been prepared including an adult skull fragment containing a left maxilla (SAM-PK-K1334; Norman et al. 2011[21]: Figs 30-33), the snout end of a subadult skull (SAM-PK-K10487; Butler 2008; Figs 38, 39), and the posterior half of another subadult skull (AMNH 24000; Figs 2C, 40-52, 61). All of these specimens except AMNH 24000 were considered in a recent more detailed description of the skull (Norman et al. 2011[21]). The comments below extend that description and discuss aspects of cranial morphology where the interpretation here (Fig. 59) differs from either their text or figures. The postcranium was originally described on the basis of a single nearly complete skeleton (SAM-PK-K1332; Santa Luca et al. 1976[4]; Santa Luca 1980[28]; Table 1). Postcranial bones originally associated with the holotype (SAM-PK-K337; Crompton and Charig 1962[1]) apparently have been lost (Norman et al. 2011[21]). Postcranial bones of a second adult individual were also collected and some of these are described below to document variation in coossification (SAM-PK-K1328; Fig. 70). Comments on the postcranium outline several anatomical areas where my interpretations differ from either the text or figures in Santa Luca (1980)[28].
Cranium
The premaxilla, best preserved in SAM-PK-K1332, exhibits several unusual features that also characterize other heterodontosaurids. The narial fossa is deep ventral to the external nares, and the fossa extends close to the alveolar margin of the premaxilla (Figs 57A, 59). The fossa was shown somewhat farther from the alveolar margin in some previous reconstructions (Norman et al. 2011[21]; Fig. 58). In Lesothosaurus (Sereno, 1991: fig. 6C), in contrast, the narial fossa is shallow ventral to the external nares, and the external nares are positioned closer to the alveolar margin.
The internarial bar is broken in all specimens preserving the end of the snout, as reported by Norman et al. (2011)[21]. The internarial bar in SAM-PK-K1332, however, was more complete as originally preserved (Fig. 90A; compare Norman et al. 2011[21]: append. 4A). Both premaxillary and nasal internarial processes extend toward one another, but their tips are broken. The internarial bar, thus, is depicted here as broken (Fig. 57A, 59). Norman et al. (2011[21]: 20) suggested the opposing internarial processes were complete and separated by a gap (Fig. 57B, 58). Maintenance of at least a slender internarial bar, as preserved in Tianyulong (Figs 9C, 22, 23), is the common condition among nonavian archosaurs. An incomplete internarial bar, on the other hand, occurs commonly in mammals in association with more extensive development of narial soft tissues (narial cartilage, snout musculature; Witmer et al. 1999[31]).
The posterolateral process of the premaxilla is quite broad at its base dorsal to the diastema in Heterodontosaurus, broader than the width between the narial fossa and anterior margin of the diastema. As a result, the posterolateral process appears somewhat swollen at its base in Heterodontosaurus, as also seen in Abrictosaurus and Tianyulong (Figs 23, 35). The posterior extremity of the posterolateral process was initially reconstructed as contacting the palpebral (Fig. 57A). Later reconstructions show it slightly shorter but inserting between the prefrontal and lacrimal (Fig. 57B) or coming to a point contact with the prefrontal (Fig. 58). The best-preserved specimen suggests the second option, that the process terminates between the lacrimal and prefrontal (Fig. 59), excluding external nasal-lacrimal contact as occurs in euornithopods.
The maxilla forms the ventral and anterodorsal borders of the triangular external antorbital fenestra (Figs 59, 90). The large invaginated opening on the snout sidewall is identified here as the external antorbital fenestra (following Witmer 1997[32]), which is backed by an inset lamina of bone. The partially enclosed recess is termed the antorbital fossa, which is bounded by the maxilla, lacrimal and jugal. Two fenestrae within the antorbital fossa include the internal antorbital fenestra (= “antorbital fenestra”, Norman et al. 2011[21]: 204), bounded by the maxilla and lacrimal, and a more anteriorly positioned opening here termed the accessory antorbital fenestra (= “anterior maxillary fenestra”, Norman et al. 2011[21]: 204), which resembles an accessory opening of similar position in Hypsilophodon (Galton, 1974a: fig. 4). The accessory antorbital opening is fully exposed only in SAM-PK-K1332, where it is positioned on the right side a little more dorsolaterally under the rim of the external antorbital fenestra as compared to the left side. The dorsal margin of the fenestra, which is isolated in the maxilla, is rounded and beveled, suggesting that the opening may have served a pneumatic function for extension of an air sac from the nasal cavity into the antorbital fossa.
In addition to these openings, there is an additional blind recess within the antorbital fossa near its anterior corner, here labeled the promaxillary fossa (Fig. 90). In its location and anterior extension, this antorbital invagination resembles the promaxillary fossa previously described among theropods (Witmer 1997[32]). Whether these structures are homologous remains an open question, as an invagination in a similar location is not known among basal sauropodomorphs such as Eoraptor (Martinez et al. 2011[33]). In computed-tomographic scans of the skull of Heterodontosaurus, Norman et al. (2011[21]: 204) observed an “opening” in the anterior corner of the antorbital fossa. This is probably the result of damage and loss after the original molding of the skull in question (SAM-PK-K1332). A blind recess is present in the anterior corner of the antorbital fossa as originally preserved (Fig. 90B).
The ventral border of the external antorbital fenestra is straight, relatively sharp-edged, and strongly everted, as in all heterodontosaurids preserving this region of the maxilla (Figs 59, 90). The rim around the anterior corner of the fenestra, by contrast, is slightly swollen. The buccal emargination extends anteriorly to the arched diastema ventral to the everted rim. Some reconstructions have depicted the ventral rim of the antorbital fossa as more rounded (Figs 57B, 58) or have shown the buccal emargination tapering toward the anterior end of the tooth row (Fig. 58) rather than extending anteriorly to the edge of the arched diastema.
The posteroventral corner of the antorbital fossa extends onto the jugal below the orbit, tapering to an end at the base of the jugal horn (Figs 57A, 59). The fossa on the jugal has been depicted previously as either considerably shorter (Fig. 57B) or longer, extending onto the jugal horn (Fig. 58). Likewise, various reconstructions have been given for the suture between the jugal and lacrimal along the orbital margin and the contribution of the jugal to the boundary of the external antorbital fenestra and wall of the antorbital fossa (Figs 57, 58). This region is best preserved in two specimens (SAM-PK-K1332, -K1334). The lacrimal-jugal suture is a scarf joint similar to that in many ornithischians; the jugal border of the external antorbital fenestra forms its arcuate posteroventral corner; and the jugal contribution to the medial wall of the antorbital fossa is limited to the extension of the fossa onto the external surface of the jugal (Fig. 59).
In dorsal view of the snout, a median fossa is present on the nasals (median “sulcus”; Norman et al. 2011[21]: 202) in both subadult and adult skulls (SAM-PK-K1332, -K10487). This fossa, bounded on each side by a rounded rim formed by the nasals, characterizes all heterodontosaurids that preserve this portion of the skull as well as several other basal ornithischians such as Agilisaurus (Peng 1992[34]). A subtriangular fossa with no apparent connection to the antorbital fossa is also present on the lateral aspect of the lacrimal (Fig. 59), a depression regarded here as a diagnostic character for Heterodontosaurus tucki.
The jugal horn has a flattened subrectangular shape and projects laterally (Fig. 59). The horn has been variously depicted as subconical (Fig. 58) or as directed ventrolaterally (Fig. 57). The postorbital has a broad fossa that excavates most of the lateral surface of the ventral and posterior rami (Fig. 59). The shape of these rami varies among heterodontosaurids. The posterior ramus in Heterodontosaurus is particularly slender (Fig. 59), although it has been reconstructed with broader proportions (Figs 57, 58).
The quadrate head is not expanded transversely in lateral view, nor is there a gap between the quadrate shaft and the squamosal and quadratojugal as sometimes reconstructed (Fig. 57). In lateral view of the skull, the ventral portion of the occipital condyle and basal tubera are visible posterior to the quadrate shaft and ventral to the paroccipital process, as preserved on the right side of SAM-PK-K1332 (Fig. 59). Some skull reconstructions have shown neither of these structures in lateral view or only the occipital condyle (Figs 57, 58).
The form of the jaw joint carried particular functional significance and is well preserved in SAM-PK-K1332 and AMNH 24000. In both specimens, an axis through the quadrate condyles angles anteromedially at about 45° as seen in ventral view (Figs 61C, 92B). In posterior view, the ventral articular surface of the quadrate condyles also angles approximately 45° ventrolaterally (Weishampel 1984[7]: fig. 3a; Fig. 92A). The articular cup, or cotylus, for the quadrate condyles is deeply concave and shaped with a snug fit, such that fore-aft (propalinal) movement of the condyles relative to the lower jaw would not have been possible. The lateral edge of the cotylus, likewise, is prominent and shaped to receive the bulbous lateral condyle of the quadrate (Fig. 61C, D). Lateral movement of the condyles relative to the lower jaw thus would not have been possible. Previous reconstructions have shown the articular cup as more broadly open or loosely fitted to the condyles in a way that might allow either fore-aft or transverse movement of the quadrate relative to the lower jaw (Figs 57, 58).
Lower jaw
The predentary is smaller relative to the anterior end of the dentary, which is expanded dorsoventrally (Figs 59-61). The predentary is triangular in lateral view, approximately as long anteroposteriorly as deep dorsoventrally, with sharp lateral margins and a rounded ventral keel. In dorsal view, the predentary is also triangular in shape and sharply pointed anteriorly. The lateral and ventral processes of the predentary are best understood only when the element is completely exposed (SAM-PK-K1332). The ventral process is the most reduced, extending as a short point in the midline that is not otherwise differentiated from the body of the predentary (Fig. 61B). The lateral processes extend posterodorsally as short prongs with a subtriangular cross-section. Their distal tips are positioned just anterior to the caniniform tooth (Figs 59, 60B). There is a deep median trough between the lateral processes, which gives the predentary a V-shaped cross-section at mid-length (Fig. 60B). Thus the predentary in Heterodontosaurus is not a solid wedge-shaped bone, but rather one with short lateral processes, a rudimentary ventral process, and a trough-shaped oral surface.
A well-defined, smooth articular surface for the predentary is present on the anterior end of the dentary, which is well preserved in SAM-PK-K1332 and SAM-PK-K10487 (Fig. 59). This surface is strongly concave anteroposteriorly and gently convex dorsoventrally (Figs 39, 59). These opposing curvatures create a smooth, vertically deep, saddle-shaped articular surface for the predentary. This surface is broadest anteroposteriorly at mid height, where the dentary projects anteriorly to a blunt end (Fig. 39). The predentary-dentary joint has been erroneously described as “spheroidal” (Weishampel 1984[7]: 47). A fragment has been lost from the anterior end of the left dentary of SAM-PK-K1332, leaving an angular breakage surface, which has been erroneously interpreted as a depression for articulation with the predentary (Weishampel 1984[7]: fig. 3f). A raised subtriangular area, here identified as the dentary boss, is present on the ventral aspect of the dentary immediately behind the predentary articulation and below the anterior dentary foramen (Figs 59, 61B).
The dentary symphysis is restricted to the ventral portion of the dentary, and is slightly thickened and rugose (Fig. 61A). It has an asymmetrical V-shape due to a small posterior embayment representing the anterior terminus of Meckel’s canal, a narrow trough near the ventral margin of the dentary ramus (Figs 56, 61A). The symphysis has been drawn and reconstructed as extending across most or all of the anterior end of the dentary (Norman et al. 2011[21]: Figs 18B, 19B) rather than limited to its ventral one-half. The more restricted, ventrally positioned symphysis may well have important functional ramifications regarding potential long-axis rotation of the dentary during mastication.
Between the symphysis and the base of the caniniform tooth is a dorsoventrally concave surface with large vascular foramen that runs anteriorly toward the predentary (Fig. 61A). This concave surface, when joined by its opposite, forms a spout-shaped trough above the symphysis that continues anteriorly onto a similar surface on the predentary (Fig. 60B). Like other ornithischians, the symphysis is located at the anterior extremity of the dentary, Meckel’s canal extends into the symphysis resulting in a V-shaped posterior embayment, and a trough-shaped surface is located above the symphysis that extends onto the predentary (Sereno 1991[35]). The ornithischian “spout-shaped” symphyseal region, thus, is not absent as reported previously (Butler et al. 2007: 19) but is proportionately narrower at least in Heterodontosaurus. The symphyseal surface between the dentaries, in addition, is more substantial than in other basal ornithischians. The symphyseal region in Heterodontosaurus, in summary, shares some features with ornithischians but appears to be less mobile. The predentary-dentary articulation, on the other hand, appears to be more mobile in advanced heterodontosaurids such as Heterodontosaurus. The symphysis in Heterodontosaurus does not resemble the plesiomorphic saurischian condition. In basal saurischians such as Eoraptor (Martinez et al. 2011[33]), the dentary rami typically approach one another at a narrow angle of incidence, and the symphyseal surface is barely differentiated from the medial aspect of the dentary.
The arched buccal emargination is deepest below the center of the dentary tooth row and extends from the base of the caniniform tooth anteriorly to the base of the coronoid process posteriorly (Figs 59, 60B). A strong coronoid process extends posterodorsally at about 45° and is overlapped medially by a coronoid bone, which is preserved in articulation in SAM-PK-K1332 and AMNH 24000 (Figs 41, 56). In basal ornithischians such as Lesothosaurus, the coronoid runs anteriorly along the alveolar margin as an elongate strap-shaped bone with a tongue-shaped posterior end that curves upward. It is only weakly developed for insertion of adductor musculature, is not visible in lateral view of the lower jaw, and has only incidental sutural contact with the surangular (Sereno 1991[35]: fig. 13G). In heterodontosaurids, in contrast, the posterior portion of the coronoid is expanded dorsally, is exposed in lateral view of the lower jaw, and is sutured to the surangular (Fig. 56, 59). The coronoid, thus, is expanded as a site of attachment for the adductor musculature, similar to the condition in basal euornithopods such as Hypsilophodon (Galton 1974a[36]: fig. 10).
A large oval depression, here termed the external mandibular fossa, is present on the angular and surangular, bordered above by a strong surangular ridge and below by the everted ventral margin of the angular (Fig. 59). The angular-surangular suture courses across the middle of the external mandibular fossa, which is present in many heterodontosaurids. There are two surangular foramina, anterior and posterior. The parallelogram-shaped anterior surangular foramen, which tends to be the larger of the pair in heterodontosaurids, is absent in the basal ornithischian Lesothosaurus but present in some euornithopods such as Hypsilophodon (Galton 1974a[36]; Sereno 1991[35]). Neurovascular grooves extend from both foramina across the surangular, the groove passing posteriorly from the anterior surangular foramen dissipating as it nears the jaw joint. These features are well preserved in a referred subadult skull (Figs 40, 41).
Norman et al. (2011)[21] erroneously described several aspects of this portion of the lower jaw due to damage and coossification in specimen SAM-PK-K1332 (Fig. 58). The angular-surangular suture was drawn just under the swollen dorsal margin of the surangular, and the anterior surangular foramen and its groove were envisioned as a fissure separating a pair of processes with “unique” articular relations with the dentary (Norman et al. 2011[21]: 187, 210). The ventral of the two processes was shown articulating laterally with an expanded dentary coronoid process (Fig. 58). The form and articular relations of the coronoid process of the dentary, surangular and angular, however, are much more conventional (Figs 40, 41, 59). The external mandibular fossa and the grooves emanating from the surangular foramina, however, may be related to more elaborate insertion of jaw musculature on the lateral aspect of the lower jaw in heterodontosaurids as discussed below.
The articulation for the quadrate is cup-shaped and fitted tightly to the lateral condyle of the quadrate. The external rim of the articular socket is everted and curled dorsally, rising to a low process immediately posterior to the posterior surangular foramen, as in Lesothosaurus and Hypsilophodon. There is one additional oval depression, here termed the lateral retroarticular fossa, which is located distal to the jaw articulation on the lateral aspect of the retroarticular process (Fig. 59). A fossa of similar form is not known elsewhere among basal ornithischians, although the condition in other heterodontosaurids remains unknown.
Premaxillary teeth
The first two premaxillary teeth (pm1, 2) are preserved in the holotypic skull (Norman et al. 2011[21]: fig. 20). The second premaxillary tooth (pm2) also was originally preserved in the referred adult skull (Figs 90A, 91) but subsequently has been lost (Norman et al. 2011[21]). The gentle swelling of the base of the crown, limited recurvature, and reduced ornamentation in pm 1 and 2 closely resemble the mesial premaxillary teeth in other basal ornithischians such as Lesothosaurus (Sereno 1991[35]). The mesial and distal carinae in these premaxillary teeth are not well exposed but appear to lack serrations as reported by Norman et al. (2011)[21]. Butler et al. (2008: 19) suggested that there is no swelling of the crown above the root in the premaxillary teeth of heterodontosaurids as occurs in other ornithischians. Although subtle, such swelling of the crown above the root is present in the mesial two premaxillary teeth in Heterodontosaurus, Echinodon, Lycorhinus and the Kayenta heterodontosaurid. Some authors (Crompton and Charig 1974; Weishampel 1984[7]; Crompton and Attridge 1986[8]; Weishampel and Witmer 1990[12]) have figured or described a distinct heel or step on the distal or lingual side of the crown (Fig. 57), but none is present (Fig. 91).
The third premaxillary crown (pm3), in contrast to the mesial two, is caniniform with markedly greater size, more pronounced recurvature, and more lateral compression of the upper crown (Figs 59, 90, 91). Basal crown width (mesiodistal) and height, however, are slightly less than the crown of the dentary caniniform tooth. Skull reconstructions showing these opposing caniniform teeth in labial view as equal in width or height are erroneous (Fig. 57). The central axis of the caniniform crown is vertical. The mesial carina is convex, whereas the distal carina is nearly straight (Figs 90A, 91). At least the distal carina is serrate, with approximately six serrations per millimeter (Norman et al. 2011[21]). The tip of the crown of pm3 is broken on both sides of the holotypic skull, with some abrasion rounding the edges of the broken surface.
Maxillary teeth
There are 11 or 12 maxillary teeth in the best-preserved adult skulls. The small broken crown base of a twelfth tooth was shown on the right side of the holotypic specimen (Crompton and Charig 1962[1]: fig. 1A), although there appears to be evidence in this specimen for only 11 maxillary teeth. Subadult skull AMNH 24000 shows that there is only one small maxillary crown at the distal end of the tooth row, as preserved on both sides (Fig. 40). On the right side of skull SAM-PK-K1332, however, it is not clear whether there are two small distal maxillary crowns or one (Fig. 55; labeled m11). A slightly larger adult skull with 12 dentary teeth does not preserve enough of the maxillary tooth row to provide a reliable count (NM QR 1788; Porro et al. 2011[20]). Variation in adult cheek tooth count seems to have been minimal in advanced heterodontosaurids such as Heterodontosaurus.
The maxillary tooth row is not straight as reconstructed in ventral view by Norman et al. (2011[21]: fig. 13). As best preserved in the adult cranium SAM-PK-K1332 (Norman et al. 2011[21]: fig. 6B), the anterior two and posteriormost tooth in the maxillary tooth row diverge labially (Fig. 60A). The ends of the maxillary tooth row, thus, curve laterally and are matched by a similar more subtle curvature of the dentary tooth row (Fig. 60B). The curvature of the cheek tooth rows has important ramifications when considering possible occlusal mechanics.
Mesiodistal crown width increases gradually from m1 to m8 and then decreases slightly in m9 and m10 and then more so in m11 (Figs 55, 59). Tooth size, thus, precisely speaking, does not increase toward the “middle of the tooth row” in Heterodontosaurus but rather distal to the center of the tooth row (contra Weishampel 1984[7]: 53), as reported by Norman et al. (2011)[21]. This pattern in crown size differential is consistent with that in other ornithischians such as Lesothosaurus, which typically have significantly smaller crowns at each end of the tooth row (Sereno 1991[35]). By comparison, crown size differential in heterodontosaurids, is enhanced by the relatively low number of crowns in the cheek dentition (Fig. 59).
The maxillary and dentary crowns in Heterodontosaurus, unlike those of other heterodontosaurids, have flattened mesial and distal surfaces that butt against one another along the tooth row, as shown in horizontal computed tomographic cross sections (Fig. 48–52). Unworn maxillary and dentary crowns are fan-shaped, gently expanding mesiodistally towards a denticulate apical margin that slopes at a low angle in mesial and distal directions away from the apical denticle (Fig. 41; crown d11). A gentle mesiodistal constriction is present between the crown and its slightly swollen, hollow root (Fig. 42). Heterodontosaurus, however, lacks the discrete cingulum or swollen shoulder present in other heterodontosaurids such as Echinodon, Lycorhinus and Abrictosaurus.
The labial surface is dominated by a strong primary ridge that is flanked by mesial and distal paracingular fossae, the former extending slightly farther toward the crown base than the latter (Fig. 41). The primary ridge, which tends to be offset toward the mesial edge of the crown face, and mesial and distal marginal ridges create a W-shaped leading edge to the wear facets (Norman et al. 2011[21]; Fig. 51). Secondary ridges extend from denticles to either side of the apical denticle. They taper to an end around mid height on the crown and are lost in heavily worn crowns. The distalmost maxillary crown appears to have more squat proportions, the secondary ridges almost reaching the base of the paracingular fossae (Fig. 41).
The lingual surface of mesial maxillary crowns is best exposed in SAM-PK-K1332, which have short primary and secondary ridges (Norman et al. 2011[21]: fig. 24C). In the largest crowns toward the distal end of the tooth row, the primary ridge is flanked by paracingular fossae as on the labial side of the crown (Fig. 50C, m10). When a wear facet obliterates most or all of the lingual side of the crown, the central fossa on the basal portion of the root creates a concave margin in cross section (Fig. 49C, m10). Maxillary crowns are also canted lingually relative to the vertical axis of their roots (Figs 43D, 44B). The enamel thins across the flat mesial and distal surfaces of the crown. Computed tomographic sections suggest that a thin layer of enamel may be present on the lingual side of the maxillary crowns, which is often obliterated by tooth wear (Figs 48–52).
In occlusal view the maxillary crowns are butted against each other, the mesial margin often slightly concave and the distal margin flat or slightly convex in cross section (Figs 51B, 52B). Although generally aligned, the wear facets in adult cheek teeth in Heterodontosaurus do not form a seamless wear surface as noted by Norman et al. (2011)[21]. They do not form a “tooth battery” (contra Norman et al. 2011[21]), the latter term reserved for tooth-supported dentitions, the roots of which are pressed against other crowns in an open alveolar trough. In Heterodontosaurus the roots of all cheek teeth are anchored within individual alveoli.
The cheek teeth in Heterodontosaurus are unusual in two other regards—their internal structure and the form of their roots. The pulp cavity of cheek teeth is spacious and extends up into the crown as is well exposed by erosion (Fig. 42) and seen in cross section (Figs 45C, 50C). In the dentition of an adult skull, the pulp cavity is exposed on wear surfaces (Fig. 55B, C). The common ornithischian condition, in contrast, is that the pulp cavity closes during maturation of the tooth, such that the pulp cavity is limited to the root (Figs 53D, 54D). The second unusual feature of the cheek teeth is the bluntly rounded distal end to the roots (Figs 42, 44B). The roots of most ornithischian teeth are more tapered toward their tips, which may retain a small distal opening (Fig. 53B, D). The functional significance of blunt-rooted cheek teeth is not known.
Dentary teeth
Unlike Echinodon and Lycorhinus, thereis no rudimentary dentary tooth preceding the caniniform tooth in Heterodontosaurus. The central axis of the dentary caniniform tooth, the largest tooth in the dentition, is tilted mesially toward the premaxillary caniniform tooth. Its root curves posteroventrally into the body of the dentary as in other heterodontosaurids (Figs 38, 59). When the lower tooth row is held horizontal, the tip of the caniniform crown is positioned over the mesial rather than distal edge of the crown base, as preserved on both sides of SAM-PK-K1332 (Norman et al. 2011[21]: Figs 16, 17). The caniniform tooth is slightly more vertically oriented in the holotypic skull, although still mesially canted (Norman et al. 2011[21]: fig. 1). In some skull reconstructions, the caniniform has been incorrectly shown with a vertical orientation (Fig. 57A). Mesial and distal carinae of the caniniform tooth are serrate. The serrations are limited to the apical one-half of the mesial margin.
There are 10 postcaniniform dentary crowns (d2-11) opposing 11 maxillary crowns. The dentary tooth row, nonetheless, terminates distally even with (SAM-PK-K1332), or distal to (AMNH 24000), the last maxillary tooth, because d1-4 crowns are relatively broader than their counterparts in the maxilla (Figs 41, 55, 59). Mesiodistal crown width increases from d2 to d8 or d9 and then decreases slightly in d10 and more dramatically in d11. Postcaniniform tooth count was initially reported as 12 or 13 (Crompton and Charig 1962[1]; Charig and Crompton 1974[30]), although only 10 postcaniniform alveoli are present in right and left dentaries in SAM-PK-K1332 (Hopson 1980[6]; Norman et al. 2011[21]; Fig. 59).
Cervical vertebrae
The vertebral column includes a well exposed cervical series starting anteriorly with the axis (Figs 62, 63). As in other vertebrae, the axial centrum and neural arch are completely coossified, suggesting that the referred skeleton (SAM-PK-K1332) pertains to a mature individual. Anteriorly, the axial intercentrum is cup-shaped and fused to the axial centrum. The wedge-shaped odontoid, likewise, is fully coossified and is beveled for articulation with the occipital condyle (Fig. 63A). The posterior face of the axial centrum is gently concave. The relatively low-angled neural spine is proportionately long, projecting farther over C3 than in most other basal ornithischians such as Lesothosaurus and Hypsilophodon (Galton 1974a[36]; Sereno 1991[35]). Unlike other ornithischians, the axial neural spine is relatively narrow, tapering in transverse width from the postzygapophyses to the posterior termination of the spine.
The parapophysis and diapophysis of cervical vertebra 3 and successive cervical vertebrae are more prominently developed than in other basal ornithischians such as Lesothosaurus (Sereno 1991[35]). The diapophysis is already developed as a subcylindrical process in C3 and the parapophysis likewise by C4 (Figs 62, 63B). The neural spine of C3 is unusually elongate for an ornithischian (Fig. 62). A hypertrophied epipophysis extends posterodorsally over the proximal one-half of C4. Joined medially for much of its length by the neural spine, this composite process resembles the axial spine in shape and orientation. Unlike the condition in the axis, however, a ridge from the prezygapophysis joins the epipophysis over the postzygapophysis (Figs 62, 63B). The basal ornithischian Lesothosaurus also has an epipophysis on C3, but it is developed only as a blunt process that does not extend beyond the postzygapophysis (Sereno 1991[35]).
The postzygapophysis of C4 bears an epipophysis that is joined by a ridge from the prezygapophysis. Although developed as a prominent process, the epipophysis does not extend beyond the postzygapophysis. The neural spine also has a broad base that may be incomplete distally (Figs 62, 63B).
The postzygapophyses of C5 and C6 are joined to the prezygapophyses by a low ridge but no epipophyseal processes are present. A marked change in the shape of the prezygapophysis occurs between C5 and C6, the latter more erect and supporting a broad hook-shaped articular surface (Figs 62, 63B). As noted by Santa Luca, the neural spines of C5 and C6 project anterodorsally between the postzygapophyses of the preceding vertebra, another condition unique among ornithischians. No articulation occurs between these spines and the postzygapophyses of the preceding vertebrae.
The length of the centra varies along the cervical column with C6-C9 noticeably shorter in length, as observed by Santa Luca (Figs 62, 63C). In this connection, the centrum of C3 is also measurably shorter than that of C2, C4 and C5. Thus, there exists the unusual condition in Heterodontosaurus in which the centra of C4 and C5 rival the axial centrum in length (Table 7). Variation in centra shape, from a parallelogram to a trapezoid, imparts an S-shaped curve to the cervical series in natural articulation (Fig. 62). The cervical centra are slightly amphicoelous with no development of opisthocoelous articulations as occurs in derived euornithopods with a highly flexed cervical series.
Santa Luca tentatively identified the transition between cervical and dorsal vertebrae, and further information supports his conclusions. The best indicator of the transition from cervical to dorsal vertebrae in ornithischians is the abrupt dorsal shift of the parapophysis from the centrum to the base of the transverse process on the neural arch. The position of the parapophysis on the anterodorsal corner of the centrum in C9 is similar to that in preceding cervical vertebrae (Figs 62, 63C). Most of this portion of D1 is exposed with no sign of a prominent parapophysis. The rib of C9, in addition, has a significantly longer tuberculum than the rib of D1, both of which are near their natural articulation. The proportions of the rib of D1 suggest that the parapophysis was located at the base of the transverse process, rather than below the neurocentral suture. Thus the evidence suggests that there are nine cervical vertebrae.
Sacral vertebrae
Santa Luca (1980)[28] identified 12 dorsal and 6 sacral vertebrae, describing S1 and S2 with coossified centra and ribs that articulate with the preacetabular process of the ilium. These centra are clearly coossified, as is exposed ventral to the left preacetabular process (Fig. 68). The vertebra he identified as S2, however, is also clearly homologous with S1 in basal dinosaurs such as Lesothosaurus, which have only five sacral vertebrae (Sereno 1991[35]; Butler 2005[37]). In Heterodontosaurus the rib of this vertebra is exposed in dorsal view as a flat strut articulating at mid-length along the preacetabular process (Fig. 68). S3, according to Santa Luca (1980)[28], has a stout sacral rib articulating with the pubic peduncle, the typical attachment for S2. The vertebrae he identified as S4-S6 have ribs or transverse processes that articulate with the iliac blade. The vertebra he identified as S6 contacts the brevis shelf at mid-length along the postacetabular process, as is typical of S5 in many other basal ornithischians (Fig. 68).
Dorsal and sacral counts hinge on the interpretation of the transverse process and rib in presacral vertebra 21 (girdle contact, not central fusion, is the arbiter regarding status as a sacral vertebra). Although the transverse process is broken at its tip, a portion of its rib is preserved projecting under the preacetabular process of the ilium (Fig. 68). An articulation with the pelvic girdle, if it existed, cannot be observed. The pelvis has undergone some transverse crushing, during which the left ilium has shifted posteroventrally. The sacral attachments that extend laterally on the right side, for example, angle posterolaterally on the left side. When restored to its original more elevated position, the left preacetabular process may well have arched over the rib of presacral vertebra 21, which is here interpreted as D13 (Fig. 68).
In most ornithischians, the end of the preacetabular process is free of sacral rib articulations. In the euornithopod Thescelosaurus neglectus (Galton 1974b[38]), however, a dorsal rib contacts with the distal end of the preacetabular process, although neither the intercentral suture nor rib-ilium articulation are coossified. Given the position and apparent brevity of the rib of presacral vertebra 21 in Heterodontosaurus, this vertebra is tentatively regarded as the last dorsal (D13). This is a low number, as most basal ornithischians have 14-16 dorsal vertebrae. The position of the rib on the right side of presacral vertebra 21 could be determined by computed tomographic imaging.
Sternum, ribs and ossified tendons
Santa Luca (1980: 173, fig. 23) mentioned the possible presence of a sternal plate, but he did not figure this bone and omitted it from his skeletal reconstruction. Subsequent skeletal reconstructions show ossified sternal plates (Bakker 1986[10]: 453, 455) or possibly ossified plates and sternal ribs (Brett-Surman 1997[14]: fig. 24.1B).
A pair of crescentic ossified sternal plates is present in SAM-PK-K1332, resembling in shape those in basal neornithischians such as Hypsilophodon (Galton 1974a[36]) or Psittacosaurus (Sereno et al. 2009[39]). Most of the left sternal plate is exposed in dorsal view posterior to the left forelimb (Fig. 64). Its lateral margin is rounded and gently concave, and its proximal end is narrow and thickened. A rounded process and notch are present along its distal edge, presumably for cartilaginous rib attachment. The medial margin, some of which is obscured by matrix, appears to be convex and longer than the lateral margin. The arcuate margin of an unossified fenestra appears to be present in the center of the left sternal plate (Fig. 64). Unlike Hypsilophodon, there are no ossified sternal ribs.
Although the axial rib is not preserved, a prominence straddling the neurocentral suture probably represents the diapophysis for a small two-headed axial rib (Fig. 62). The rib of C4 is preserved near its natural articulation and closely resembles that in Hypsilophodon (Galton (1974a)[36]. The capitulum is shorter and stouter than the tuberculum and more closely associated with a low spinous process. The rib shaft would have extended posteriorly just beyond the posterior edge of the centrum (Fig. 62). The rib of C5 is similar, the proximal processes Y-shaped and more divergent. Most of the ribs of C8 and 9 are exposed near the left scapulocoracoid. These are robust, long ribs with a longer capitulum and tuberculum then in Hypsilophodon (Galton 1974a[36]).
Ossified tendons are present from the anterior dorsal through the sacral vertebrae (Figs 62, 68), a distribution that may be primitive for ornithischians. The first trace of ossified tendons occurs just posterior to the neural spine of D4, and the last trace is present on S4. All are preserved as slender, non-bifurcating, floating epaxial rods that are located between the transverse process and neural spine.
Forelimb
The reconstruction of the forearm presented by Santa Luca (1980: Figs 13, 15) did not remove postmortem fracturing and movement that is visible in the right forearm. A fracture crosses the mid-shafts of the right radius and ulna (Fig. 65). The portion of these bones proximal to the fracture has rotated about 30°, so that the olecranon process is exposed in profile and the flattened end of the ulna lies parallel to a plane through the carpus and metacarpus (Fig. 65). The left ulna, in contrast, shows an uncrushed condition, in which a plane through the coronoid and olecranon processes of the ulna is canted at about 45° to the plane of the carpus and metacarpus. In the reconstruction presented here, the proximal portions of the radius and ulna are restored to their natural orientation (Fig. 67A).
Santa Luca reconstructed the metacarpus with the base of metacarpal 2 inset into the carpus relative to metacarpal 1, such that its base articulates medially with distal carpal 1 (Santa Luca 1980[28]: fig. 13; Langer and Benton 2006[16]: fig. 8A). This unusual condition, which is unknown elsewhere in dinosaurs, should not be confused with the basal sauropodomorph condition, in which metacarpal 1, rather than metacarpal 2, is inset into the carpus and articulates laterally with distal carpal 2 (Sereno 2007[40]). Santa Luca’s interpretation was based on the right carpus and manus, which shows this configuration (Fig. 65). In the left forelimb, however, the bases of metacarpals 1 and 2 are aligned (Fig. 64), their squared bases in mutual and matching contact. The apparent inset of metacarpal 2 in Heterodontosaurus, thus, appears to be an artifact of preservation in the right forelimb. The bases of metacarpals 1 and 2 were probably in alignment (Fig. 67C, as also appears to be the case in the largely articulated manus of Abrictosaurus (Fig. 36). The base of metacarpal 3, in contrast, may have been positioned slightly distal to metacarpal 2, as this is preserved on both right and left sides in Heterodontosaurus (Fig. 65) and in Abrictosaurus (Fig. 36).
Metacarpals 1-4 have squared bases that abut one another (Figs 65, 66, 67C), a striking similarity to the condition seen in primitive theropods such as Herrerasaurus (Sereno 1993[41]) and Eodromaeus (Martinez et al. 2011[33]). Abrictosaurus (Fig. 36) and the Kayenta heterodontosaurid appear to show a similar metacarpal condition, which may characterize heterodontosaurids in general.
Bakker and Galton (1974[9]: fig. 1H) figured the manus of Heterodontosaurus with strong rotation in digit I. They showed the ungual of digit I directed medially in an extended manus, the medial aspect of the ungual fully exposed in dorsal view of the manus. These authors and Bakker (1986[10]: 453) claimed that a medially twisted thumb characterizes both Heterodontosaurus and basal sauropodomorphs such as Ammosaurus (= Anchisaurus), which has strong medial rotation in the first phalanx of digit I (Sereno 2007[40]).
The right manus as preserved in Heterodontosaurus, however, shows only very slight deflection of the distal end of digit I toward the medial side (Fig. 65). This is the original preserved orientation of digit I of the right manus. Despite some damage and loss of the mid section of the first phalanx, metacarpal 1, the base of phalanx I-1 and a portion of the ungual are preserved in situ embedded in matrix (Fig. 65). The left manus, in contrast, is transversely compressed with the digits lying subparallel to one another (Figs 64, 66). Although less informative with regard to digital orientation, left phalanx I-1 is compete and preserves a nearly straight shaft (Fig. 64). That matches the preserved position of the proximal and distal ends of right phalanx I-1 (Fig. 65). Thus there is no evidence for medial rotation of the pollex in Heterodontosaurus akin to that present in Eoraptor (Martinez et al. 2011[33]) and other basal sauropodomorphs (Sereno 2007[40]).
The distal end of metacarpal 1, which is best preserved on the right side, shows asymmetric distal condyles very similar to the condition present in many theropods. The lateral condyle is larger and raised above the medial condyle (Figs 64, 67A), which as noted by Santa Luca would cause the pollex to converge toward the palm during flexion, opposite the direction shown by Galton and Bakker (1974). It is common in the pollex of theropods such as Eodromaeus and Herrerasaurus (Sereno 1993[41]; Martinez et al. 2011[33]) that the first phalanx shows a slight twist in the opposite direction. A subtle opposite twist is apparent in the first phalanx on both right and left sides in Heterodontosaurus, when comparing vertical planes through the proximal and distal ends (Fig. 64). As preserved, all of the principal inner digits in the partially flexed right manus are canted slightly medially toward their distal ends. This is probably the result of subtle postmortem compression of the manus. Given the typical form of the distal condyles of metacarpal 1, the pollex is reconstructed in neutral pose in a manner that would result in its palmar deflection upon flexion (Fig. 67A).
Hindlimb
In SAM-PK-K1332 the tibia, fibula, astragalus and calcaneum are coossified as a tibiotarsus as noted by Santa Luca. That coossification, however, is not as complete and homogeneous as in avians. The distal suture between the fibula and calcaneum, for example, is visible, and the distal end of the fibula is expanded anteroposteriorly and transversely (Fig. 69). In a second individual of similar size collected from the same formation (SAM-PK-K1328), partial sutures between the fibula, calcaneum and astragalus are visible (Fig. 70). The astragalus has a short ascending process that may establish a contact with the distal end of the fibula, and the calcaneum has a transverse width approximately 25% of the width of the proximal tarsus (Fig. 70).
Santa Luca described three distal tarsals. This would be an unusual condition for an ornithischian, which usually retain two distal tarsals interpreted as distal tarsals 3 and 4. The usual pair of distal tarsals, however, are present, the flatter broader medial distal tarsal split by a crack in the block containing the right distal tarsus and pes (Figs 67B, 71). The left tarsometatarsus confirms the presence of only two distal tarsals.
Distal tarsal 3 is partially coossified with the metatarsus, the suture between its thinner medial portion and metatarsal 1 obliterated medially and posteriorly. The cuboid distal tarsal 4 is partially coossified with the medial distal tarsal and the proximal end of metatarsal 4. It is slightly wasted with an everted dorsal rim and a foramen on its anterior face (Figs 67B, 71).
The proximal end of metatarsal 1 is reduced proximally to a thin narrow splint (Figs 67B, 71). Compared to the proximal end of metatarsal 2, metatarsal 1 has approximately 20% of its transverse width and 30% of its anteroposterior depth. Santa Luca erroneously figured the proximal end of metatarsal 1 as roughly as broad as metatarsal 2. Metatarsal 1 extends proximally to contact the tarsus, where it fuses with the medial distal tarsal. Metatarsal 5, a dorsoventrally flattened sinuous splint, is preserved in articulation on the posterior aspect of the right tarsometatarsus (Table 9). Although Santa Luca described metatarsal 5 as positioned ventral to the proximal end of metatarsal 4, it preserves articular relations seen in other ornithischians and appears to be in natural articulation, along with all other elements of the right tarsometatarsus. Its proximal end articulates with the beveled posterior face of distal tarsal 4, and its shaft angles at about 45° mediodistally across metatarsal 4 to the ventral aspect of metatarsal 3.
Table 7. Measurements (mm) of the skull and axial column of the holotypic skeleton of the South African heterodontosaurid Heterodontosaurus tucki (SAM-PK-K1332). Except for the ninth cervical centrum and the chevrons, measurements are from Santa Luca (1984). Parentheses indicate estimated measurement.
| Region |
Measurement |
|
| Cranium |
Length, anterior tip to occipital condyle |
115
|
| Height, parietal to quadrate condyle |
66
|
| Cervical vertebrae |
C2 centrum length |
16
|
| C3 centrum length |
14
|
| C4 centrum length |
16
|
| C5 centrum length |
16
|
| C6 centrum length |
13
|
| C7 centrum length |
13
|
| C8 centrum length |
13
|
| C9 centrum length |
(13)
|
| Dorsal vertebrae |
D1 centrum length |
(13)
|
| D2 centrum length |
13
|
| D3 centrum length |
14
|
| D4 centrum length |
(13)
|
| D5 centrum length |
15
|
| D6 centrum length |
15
|
| D7 centrum length |
15
|
| D8 centrum length |
15
|
| D9 centrum length |
15
|
| D10 centrum length |
—
|
| D11 centrum length |
(15)
|
| D12 centrum length |
15
|
| Sacral vertebrae |
S1 centrum length |
14
|
| S2 centrum length |
(13)
|
| S3 centrum length |
—
|
| S4 centrum length |
—
|
| S5 centrum length |
—
|
| S6 centrum length |
14
|
| Caudal vertebrae |
CA1 centrum length |
14
|
| CA2 centrum length |
—
|
| CA3 centrum length |
15
|
| CA4 centrum length |
(15)
|
| CA5 centrum length |
16
|
| CA6 centrum length |
16
|
| CA7 centrum length |
16
|
| CA8 centrum length |
17
|
| CA9 centrum length |
18
|
| CA10 centrum length |
18
|
| |
CA11 centrum length |
18
|
| CA12 centrum length |
—
|
| ?CA19 centrum length |
16
|
| ?CA20 centrum length |
16
|
| ?CA21 centrum length |
16
|
| ?CA22 centrum length |
17
|
| ?CA23 centrum length |
16
|
| ?CA24 centrum length |
17
|
| ?CA25 centrum length |
16
|
| ?CA26 centrum length |
16
|
| ?CA27 centrum length |
16
|
| ?CA28 centrum length |
17
|
| ?CA29 centrum length |
16
|
| ?CA30 centrum length |
16
|
| ?CA31 centrum length |
—
|
| ?CA32 centrum length |
16
|
| ?CA33 centrum length |
16
|
| ?CA34 centrum length |
—
|
| Chevrons |
Ch1 length |
23
|
| Ch2 length |
23
|
| Ch3 length |
22
|
| Ch4 length |
23
|
| Ch5 length |
23
|
| Ch6 length |
24
|
| Ch7 length |
24
|
| Ch8 length |
24
|
| Ch9 length |
22
|
| Ch10 length |
17
|
| Ch11 length |
15
|
| ?Ch20 length |
19
|
| ?Ch21 length |
20
|
| ?Ch22 length |
19
|
| ?Ch23 length |
17
|
| ?Ch24 length |
18
|
| ?Ch25 length |
15
|
| ?Ch26 length |
15
|
| ?Ch27 length |
17
|
| ?Ch28 length |
16
|
| ?Ch29 length |
15
|
| ?Ch30 length |
15
|
| ?Ch31 length |
16
|
Table 8. Measurements (mm) of the pectoral girdle and forelimb bones of the holotypic skeleton of the South African heterodontosaurid Heterodontosaurus tucki (SAM-PK-K1332). Except for the bones of manual digit V, measurements are from Santa Luca (1984), with right and left sides averaged when both are available. Parentheses indicate estimated measurement.
| Structure |
Measurement |
|
| Scapula |
Blade length |
86
|
| Blade, minimum width of neck |
8
|
| Blade, maximum width at distal end |
22
|
| Humerus |
Length |
83
|
| Proximal end, maximum width |
21
|
| Deltopectoral crest length |
35
|
| Shaft, minimum diameter |
7
|
| Distal end, maximum width |
19
|
| Ulna |
Length |
68
|
| Shaft, minimum diameter |
4
|
| Radius |
Length |
58
|
| Shaft, minimum diameter |
4
|
| Manual digit I |
Metacarpal 1 length |
18
|
| Phalanx I-1 length |
17
|
| Ungual I-2 length |
17
|
| Manual digit II |
Metacarpal 2 length |
23
|
| Phalanx II-1 length |
15
|
| Phalanx II-2 length |
17
|
| Ungual II-3 length |
18
|
| Manual digit III |
Metacarpal 3 length |
22
|
| Phalanx III-1 length |
14
|
| Phalanx III-2 length |
12
|
| Phalanx III-3 length |
15
|
| Ungual III-4 length |
17
|
| Manualdigit IV |
Metacarpal 4 length |
15
|
| Phalanx IV-1 length |
7
|
| Phalanx IV-2 length |
5
|
| Manual digit V |
Metacarpal 5 length |
7
|
| Phalanx V-1 length |
6
|
| Phalanx V-2 length |
4
|
Table 9. Measurements (mm) of the pelvic girdle and hind limb bones of the holotypic skeleton of the South African heterodontosaurid Heterodontosaurus tucki (SAM-PK-K1332). Except for metatarsal 5, measurements are from Santa Luca (1984), with right and left sides averaged when both are available.
| Structure |
Measurement |
|
| Ilium |
Blade length |
97
|
| Blade, height dorsal to acetabular rim |
15
|
| Preacetabular process length |
46
|
| Postacetabular process length |
24
|
| Femur |
Length |
112
|
| Minimum shaft diameter |
9
|
| Head to distal end of fourth trochanter |
46
|
| Tibiotarsus |
Length |
145
|
| Minimum shaft diameter |
9
|
| Pedal digit I |
Metatarsal 1 length |
38
|
| Phalanx I-1 length |
17
|
| Ungual I-2 length |
18
|
| Pedal digit II |
Metatarsal 2 length |
59
|
| Phalanx II-1 length |
19
|
| Phalanx II-2 length |
16
|
| Ungual II-3 length |
21
|
| Pedal digit III |
Metatarsal 3 length |
68
|
| Phalanx III-1 length |
22
|
| Phalanx III-2 length |
16
|
| Phalanx III-3 length |
14
|
| Ungual III-4 length |
18
|
| Pedal digit IV |
Metatarsal 4 length |
61
|
| Phalanx IV-1 length |
17
|
| Phalanx IV-2 length |
12
|
| Phalanx IV-3 length |
11
|
| Phalanx IV-4 length |
10
|
| Ungual IV-5 length |
16
|
| Pedal digit V |
Metatarsal 5 |
15
|
Taxon Treatment
- Sereno, P; 2012: Taxonomy, morphology, masticatory function and phylogeny of heterodontosaurid dinosaurs ZooKeys, 226: 1-225. doi
Images
| Figure 2. Early heterodontosaurid discoveries. A Lithographic drawing of the right and left premaxillae and the anterior portion of the left maxilla in lateral view of Echinodon becklesii (NHMUK 48209; from Owen 1861 [42]) B Drawing of lower jaws in dorsal view of Geranosaurus atavus (SAM-PK-K1871; from Broom 1911 [43]) C Photograph of the posterior portion of a subadult skull in right lateral view of Heterodontosaurus tucki (AMNH 24000). Scale bars equal 1 cm in A and 2 cm in B and C. |
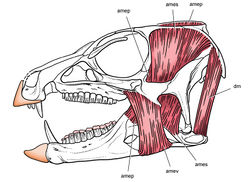
| Figure 9. More recent heterodontosaurid discoveries from northern locales. A Jaws of Fruitadens haagarorum from the Upper Jurassic Morrison Formation in Colorado, USA (based on LACM 115747, 128258; reversed from Butler et al. 2010 [44]) B Left dentary in lateral view of an undescribed heterodontosaurid from the Lower Jurassic Kayenta Formation of Arizona (from Sereno et al. unpublished) C Partial skull of Tianyulong confuciusi from the Yixian Formation of Liaoning Province, PRC (STMN 26-3; reversed from Zheng et al. 2009 [45]). Abbreviations: a angular ad 9, 10 alveolus for dentary tooth 9, 10 adf anterior dentary foramen antfo antorbital fossa apd articular surface for the predentary d dentary d1, 2, 8 dentary tooth 1, 2, 8 emf external mandibular fenestra en external nares j jugal l lacrimal m maxilla n nasal pd predentary pf prefrontal pm premaxilla po postorbital q quadrate qj quadratojugal sa surangular. Scale bar equals 1 cm in A and C and 5 mm in B. |
| Figure 22. Anterior portion of skull of the heterodontosaurid Tianyulong confuciusi from the Lower Cretaceous Jehol Group of China. Snout in right lateral view (IVPP V17090). Scale bar equals 5 mm. |
| Figure 23. Anterior portion of skull of the heterodontosaurid Tianyulong confuciusi from the Lower Cretaceous Jehol Group of China. Snout in right lateral view (IVPP V17090). Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bar equals 5 mm. Abbreviations: ad1 alveolus of dentary tooth 1 antfe antorbital fenestra be buccal emargination d dentary d1, 5, 9 dentary tooth 1, 5, 9 en external naris fo foramen l left m maxilla m4, 9 maxillary tooth 4, 9 n nasal nf narial fossa pd predentary pm premaxilla pm1, 2 premaxillary tooth 1, 2 r right. |
| Figure 35. Skull of Abrictosaurus consors from the Lower Jurassic Upper Elliot Formation of South Africa. Skull reconstruction in left lateral view based on NHMUK RU B54. Dashed lines indicate estimated edges and sutures. Abbreviations: a angular adf anterior dentary foramen antfo antorbital fossa apd articular surface for the predentary apmf anterior premaxillary foramen be buccal emargination d dentary d1, d14 dentary tooth 1, 14 de dentary expansion en external nares j jugal l lacrimal m maxilla m14 maxillary tooth 14 nf narial fossa pap palpebral pd predentary pm premaxilla pm1 premaxillary tooth 1 po postorbital q quadrate qj quadratojugal sa surangular. |
| Figure 36. Forelimb of Abrictosaurus consors from the Lower Jurassic Upper Elliot Formation of South Africa. Stereopair ( A) and line drawing ( B) of the right forelimb in anterior and ventral views (NHMUK RU B54). Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bars equal 2 cm in A and B. Abbreviations: dc distal carpal dpc deltopectoral crest fr fragment h humerus mc1-5 metacarpals 1-5 ph I-1 phalanx 1 of manual digit I ph II-1 phalanx 1 of manual digit II ph III-1 phalanx 1 of manual digit III ph III-2 phalanx 2 of manual digit III ph V-1 phalanx 1 of manual digit V po postorbital ra radius sk skull ul ulna ule ulnare. |
| Figure 38. Snout of the heterodontosaurid Heterodontosaurus tucki from from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Anterior one-half of a juvenile skull (SAM-PK-K10487). Stereopair (A) and line drawing (B) in anterolateral view. Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bars equal 1 cm in A and B. Abbreviations: apd articular surface for predentary apmf anterior premaxillary foramen d dentary d1 dentary tooth 1 f frontal fo foramen j jugal l lacrimal m maxilla mt maxillary teeth n nasal nd nasal depression nf narial fossa pap palpebral pd predentary pm premaxilla po postorbital prf prefrontal. |
| Figure 39. Snout end of Heterodontosaurus tucki from from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Close-up view of the anterior end of a juvenile skull (SAM-PK-K10487). Stereopair (A) and line drawing (B) in left lateral view. Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bars equal 5 mm in A and B. Abbreviations: apd articular surface for the predentary apmf anterior premaxillary foramen d dentary d1 dentary tooth 1 fo, foramen gr groove m maxilla mt maxillary teeth nf narial fossa pd predentary pm premaxilla. |
| Figure 40. Posterior end of the skull of Heterodontosaurus tucki from from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Posterior portion of a juvenile skull (AMNH 24000). Stereopair (A) and line drawing (B) in right lateral view. Hatching indicates broken bone; dashed lines indicate estimated edges; grey tone indicates matrix; pink tone indicates wear facets. Scale bars equal 1 cm in A and B. Abbreviations: aa articular surface for angular aqj articular surface for quadratojugal asaf anterior surangular foramen d dentary d7, 11 dentary tooth 7, 11 j jugal jfl jugal flange l left m maxilla m8, 11 maxillary tooth 8, 11 psaf posterior surangular foramen q quadrate qj quadratojugal r right sa surangular wf wear facet. |
| Figure 41. Posterior dentition of Heterodontosaurus tucki from from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Tooth wear and replacement in posterior maxillary and dentary teeth of a juvenile skull (AMNH 24000). Stereopair (A) and line drawing (B) in right lateral view. Hatching indicates broken bone; dashed lines indicate estimated edges; grey tone indicates matrix; pink tone indicates wear facets. Scale bars equal 2 cm in A and 1 cm in B. Abbreviations: asaf anterior surangular foramen c coronoid cp coronoid process d dentary d7, 11 dentary tooth 7, 11 ec ectopterygoid jfl jugal flange m maxilla m8, 11 maxillary tooth 8, 11 mpcf mesial paracingular fossa pri primary ridge sa surangular wf wear facet. |
| Figure 42. Posterior dentition of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Tooth wear and replacement in posterior maxillary and dentary teeth in a juvenile skull (AMNH 24000). Stereopair (A) and line drawing (B) in left lateral view. Hatching indicates broken bone; grey tone indicates matrix; pink tone indicates wear facets. Scale bars equal 5 mm in A and B. Abbreviations: d dentary d8-10 dentary tooth 8–10 m maxilla m10, 11 maxillary tooth 10, 11 pc pulp cavity pri primary ridge ri ridge wf wear facet. |
| Figure 43. Tooth structure, occlusion, and replacement in Heterodontosaurus tucki from the Lower Jurassic Elliot and Clarens Formations of South Africa. Successive coronal computed-tomographic sections in cutaway view of a subadult skull (AMNH 24000). A Posterior portion of skull in right lateral view showing the location of coronal cross-sections B Cross-section through maxillary tooth 9 and dentary tooth 7 C, D Cross-sections through maxillary tooth 9 and dentary tooth 8. Abbreviations: d7-9 dentary teeth 7-9 m9, 10 maxillary tooth 9, 10 ocp occlusal plane pc pulp cavity rm9 replacement maxillary tooth 9. |
| Figure 44. Tooth structure, occlusion, and replacement in Heterodontosaurus tucki from the Lower Jurassic Elliot and Clarens Formations of South Africa.Successive coronal computed-tomographic sections in cutaway view of a subadult skull (AMNH 24000). A Posterior portion of skull in right lateral view showing the location of coronal cross-sections B Cross-section through maxillary tooth 10 and dentary tooth 9 C Cross-section through dentary tooth 11. Abbreviations: d dentary d9, 11 dentary tooth 9, 11 iac inferior alveolar canal j jugal m maxilla m10 maxillary tooth 10 ocp occlusal plane r replacement tooth rd11 replacement dentary tooth 11 sac superior alveolar canal sp splenial. |
| Figure 45. Tooth structure, occlusion, and replacement in Heterodontosaurus tucki from the Lower Jurassic Elliot and Clarens Formations of South Africa.Successive sagittal computed-tomographic sections in cutaway view of a subadult skull (AMNH 24000). A Posterior portion of skull in anterior view showing the location of sagittal cross-sections B, C Cross-sections in right lateral view through right maxillary and dentary rami. Abbreviations: pc pulp cavity rd11 replacement dentary tooth 11 rm11 replacement maxillary tooth 11 sac superior alveolar canal. |
| Figure 46. Tooth structure, occlusion, and replacement in Heterodontosaurus tucki from the Lower Jurassic Elliot and Clarens Formations of South Africa. Successive sagittal computed-tomographic sections in cutaway view of a subadult skull (AMNH 24000). A Posterior portion of skull in anterior view showing the location of sagittal cross-sections B Cross-section in right lateral view through right maxillary and dentary rami C Cross-section in right lateral view through left maxillary and dentary rami. Abbreviations: psaf posterior surangular foramen q quadrate qj quadratojugal rd8 replacement dentary tooth 8 rm9, 10 replacement maxillary teeth 9, 10. |
| Figure 47. Tooth structure, occlusion, and replacement in Heterodontosaurus tucki from the Lower Jurassic Elliot and Clarens Formations of South Africa. Successive sagittal computed-tomographic sections in cutaway view of a subadult skull (AMNH 24000). A Posterior portion of skull in anterior view showing the location of sagittal cross-sections B, C Cross-sections in right lateral view through left maxillary and dentary rami. Abbreviations: pc pulp cavity q quadrate rd10 replacement dentary tooth 10 rm11 replacement maxillary tooth 11. |
| Figure 48. Tooth structure, occlusion, and replacement in Heterodontosaurus tucki from the Lower Jurassic Elliot and Clarens Formations of South Africa.Successive horizontal computed-tomographic sections in cutaway view of a subadult skull (AMNH 24000). A Posterior portion of skull in right lateral view showing the location of horizontal cross-sections B, C Cross-sections (anterior toward top of page) through the maxilla. Abbreviations: j jugal m10, 11 maxillary tooth 10, 11 q quadrate qj quadratojugal rm9, 11 replacement maxillary tooth 9, 11. |
| Figure 49. Tooth structure, occlusion, and replacement in Heterodontosaurus tucki from the Lower Jurassic Elliot and Clarens Formations of South Africa. Successive horizontal computed-tomographic sections in cutaway view of a subadult skull (AMNH 24000). A Posterior portion of skull in right lateral view showing the location of horizontal cross-sections B Cross-section (anterior toward top of page) through the maxilla C Cross-section (anterior toward top of page) through occluding portions of maxillary tooth rows.Abbreviations: m8, 10, 11 maxillary tooth 8, 10, 11 pc pulp cavity q quadrate rm9, 11 replacement maxillary tooth 9, 11. |
| Figure 50. Tooth structure, occlusion, and replacement in Heterodontosaurus tucki from the Lower Jurassic Elliot and Clarens Formations of South Africa.Successive horizontal computed-tomographic sections in cutaway view of a subadult skull (AMNH 24000). A Posterior portion of skull in right lateral view showing the location of horizontal cross-sections B, C Cross-sections (anterior toward top of page) through occluding portions of maxillary and dentary tooth rows.Abbreviations: d9, 10, dentary tooth 9, 10 m8-11 maxillary teeth 8-11 ocp occlusal plane q quadrate rm11 replacement maxillary tooth 11. |
| Figure 51. Tooth structure, occlusion, and replacement in Heterodontosaurus tucki from the Lower Jurassic Elliot and Clarens Formations of South Africa. Successive horizontal computed-tomographic sections in cutaway view of a subadult skull (AMNH 24000). A Posterior portion of skull in right lateral view showing the location of horizontal cross-sections B, C Cross-sections (anterior toward top of page) through occluding portions of maxillary and dentary tooth rows.Abbreviations: d dentary d8-11 dentary teeth 8-11 m maxilla m9, 10 maxillary tooth 9, 10 ocp occlusal plane rd10 replacement dentary tooth 10. |
| Figure 52. Tooth structure, occlusion, and replacement in Heterodontosaurus tucki from the Lower Jurassic Elliot and Clarens Formations of South Africa. Successive horizontal computed-tomographic sections in cutaway view of a subadult skull (AMNH 24000). A Posterior portion of skull in right lateral view showing the location of horizontal cross-sections B, C Cross-sections (anterior toward top of page) through the dentary. Abbreviations: d8, 9, 11 dentary tooth 8, 9, 11 rd10, 11 replacement dentary tooth 10, 11 sp splenial. |
| Figure 53. Tooth structure in Ouranosaurus nigeriensis from the mid-Cretaceous Elrhaz Formation of Niger. Successive coronal computed-tomographic sections in cutaway view of a worn maxillary tooth showing internal structure (MNBH GAD28) A Maxillary tooth in labial view showing the location of coronal cross-sections B Maxillary tooth in distal view showing the division between crown and root and the fossa for an adjacent replacement tooth C Section through mesial portion of crown D Section through mid section of crown. Abbreviations: cpc collapsed pulp cavity cr crown den dentine dent denticle e enamel es erosional surface frt fossa for replacement tooth pc pulp cavity pri primary ridge rt root wf wear facet. |
| Figure 54. Tooth structure in Ouranosaurus nigeriensis from the mid-Cretaceous Elrhaz Formation of Niger. Successive horizontal computed-tomographic sections in cutaway view of a worn maxillary tooth showing internal structure (MNBH GAD28) A Maxillary tooth in distal view showing the location of horizontal cross-sections B Maxillary tooth in apical view (labial toward top of page) C Cross-section (labial toward top of page) through mid crown D Cross-section (labial toward top of page) through base of the crown E Cross-section (labial toward top of page) through proximal portion of the root. Abbreviations: cpc collapsed pulp cavity den dentine e enamel pc pulp cavity pri primary ridge rt root sri secondary ridge wf wear facet. |
| Figure 55. Posterior dentition of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Posterior half of worn maxillary and dentary tooth row in an adult skull in right lateral view (SAM-PK-K1332). Stereopair (A) of right posterior dentary tooth row tipped labially (laterally) exposing the wear facets in dorsolateral view. Stereopair (B) and line drawing (C) of the posterior half of the tooth rows in natural articulation in lateral view. Hatching indicates broken bone; grey tone indicates matrix; pink tone indicates wear facets; yellow tone indicates accessory wear surfaces. Scale bars equal 1 cm in A and B, 5 mm in C. Abbreviations: aws accessory wear surface be buccal emargination d dentary d5, 8, 11 dentary tooth 5, 8, 11 m maxilla m5, 8, 11 maxillary tooth 5, 8, 11 pc pulp cavity wf wear facet. |
| Figure 56. Postcaniniform dentary tooth row of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Worn right dentary tooth row in an adult skull (SAM-PK-K1332). Stereopair (A) and line drawing (B) in medial view. Dashed lines indicate estimated edges; tone indicates matrix. Scale bars equal 1 cm in A and B. Abbreviations: c coronoid d dentary d2, 8, 11 dentary tooth 2, 8, 11 fo foramen imf internal mandibular fenestra; Mc Meckel’s canal sp splenial. |
| Figure 57. Skull of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Previous skull reconstructions in left lateral view A From Charig and Crompton (1974) [30] B Reversed from Weishampel (1984) [7]. |
| Figure 58. Skull of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Skull reconstruction in left lateral view from Norman et al. (2011) [21]. Abbreviations: a angular antfe antorbital fenestra antfo antorbital fossa ar articular bo basioccipital c coronoid d dentary f frontal j jugal jfl jugal flange jh jugal horn l lacrimal m maxilla n nasal nf narial fossa p parietal pap palpebral pd predentary pm premaxilla po postorbital popr paroccipital process prf prefrontal q quadrate qf quadrate foramen qj quadratojugal sa surangular sq squamosal. |
| Figure 59. Skull of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. New skull reconstruction in lateral view showing the dentition with intermediate wear (scleral ring not shown). Pink tone indicates wear facets. Abbreviations: a angular aantfe accessory antorbital fenestra adf anterior dentary foramen adi arched diastema antfo antorbital fossa apd articular surface for the predentary apmf anterior premaxillary foramen asaf anterior surangular foramen be buccal emargination bo basioccipital bt basal tubera c coronoid d dentary d2, d11 dentary tooth 2, 11 dbo dentary boss eantfe external antorbital fenestra emf external mandibular fenestra emfo external mandibular fossa en external naris f frontal iantfe internal antorbital fenestra j jugal jfl jugal flange jh jugal horn l lacrimal lrfo lateral retroarticular fossa m maxilla m1, 11 maxillary tooth 1, 11 n nasal nf narial fossa p parietal pap palpebral pd predentary pm premaxilla pm1, 3 premaxillary tooth 1, 3 pmfo promaxillary fossa po postorbital pof postorbital fossa popr paroccipital process prf prefrontal psaf posterior surangular foramen q quadrate qf quadrate foramen qj quadratojugal sa surangular sar surangular ridge sq squamosal. |
| Figure 60. Jaws of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Reconstruction of upper and lower jaws showing the dentition with heavy wear (based on SAM-PK-K1332). Dashes indicate estimated edges; pink tone indicates wear facets A Upper dentition and anterior palate in ventral view B Lower dentition, dentary symphysis and predentary in dorsal view. Abbreviations: adi arched diastema be buccal emargination d dentary d1, 2, 11 dentary tooth 1, 2, 11 fo foramen m maxilla m1, 11 maxillary tooth 1, 11 pd predentary pm premaxilla pm1, 3 premaxillary tooth 1, 3 spl secondary palate sym symphysis symt symphyseal trough v vomer. |
| Figure 61. Jaw joints of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Dentary symphysis and quadrate-articular jaw joint A Reconstruction of the anterior portion of the right dentary in medial view (based on SAM-PK-K1332) B Reconstruction of the anterior end of the lower jaws in ventral view (based on SAM-PK-K1332) C Stereopair of the right lower jaw joint in lateral view (the lateral edge of the quadrate condyle and articular are broken away exposing the tight jaw articulation) in a subadult skull (AMNH 24000) D Stereopair of the left lower jaw joint in lateral view (the lateral edge of the quadrate condyle is broken away exposing the inclined trough of the jaw articulation) in a subadult skull (AMNH 24000). Scale bar equals 1 cm in C and D. Abbreviations: d dentary d1, 2 dentary tooth 1, 2 dbo dentary boss fo foramen Mc Meckel’s canal pd predentary sym symphysis symt symphyseal trough vp ventral process. |
| Figure 62. Presacral vertebrae of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Cervical and anterior dorsal vertebrae and ribs of an adult skeleton (SAM-PK-K1332). Stereopair (A) and line drawing (B) in left lateral view. Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bars equal 5 cm in A and 3 cm in B. Abbreviations: ax axial axi axial intercentrum C2-6, 8, 9 cervical vertebra 2–6, 8, 9 D1, 3, 4 dorsal vertebra 1, 3, 4 di diapophysis epi epipophysis h humerus ns neural spine od odontoid ot ossified tendons pa parapophysis prz prezygapohpysis r rib sc scapula. |
| Figure 63. Presacral vertebrae of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Cervical and anterior dorsal vertebrae and ribs of an adult skeleton (SAM-PK-K1332). Stereopairs of C1-3 (A), C4-6 (B), and C7-D4 (C) in left lateral view. Scale bars equal 2 cm in A and B, 3 cm in C. |
| Figure 64. Pectoral girdle and forelimb of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Pectoral girdle and forelimb of an adult skeleton (SAM-PK-K1332). Photograph (A) and line drawing (B) of the sternal plates and left forelimb in dorsal and medial view, respectively. Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bar equals 3 cm. Abbreviations: I, II digit I, II ar articular surface for a sternal rib C9 cervical 9 dc2 distal carpal 2 dpc deltopectoral crest fen fenestra h humerus hd head l left mc1, 2 metacarpal 1, 2 ph phalanx r rib or right ra radius rae radiale stp sternal plate ul ulna ule ulnare un ungual. |
| Figure 65. Forelimb of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Antebrachium, carpus and manus of an adult skeleton (SAM-PK-K1332). Photograph (A) and line drawing (B) of the right radius, ulna, carpus and manus in dorsal view. Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bar equals 3 cm. Abbreviations: I-IV digits I-IV ce centrale dc1, 4 distal carpal 1, 4 ded dorsal extensor depression dip dorsal intercondylar process he heel mc1, 4 metacarpal 1, 4 ol olecranon ph, phalanx pi pisiform ra radius rae radiale ul ulna ule ulnare un ungual. |
| Figure 66. Carpus and manus of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Carpus and manus of an adult skeleton (SAM-PK-K1332). Photograph (A) and line drawing (B) of the left carpus and manus in lateral view. Hatching indicates broken bone; tone indicates matrix. Scale bar equals 2 cm. Abbreviations: II-V digits II-V dc4, 5 distal carpal 4, 5 mc3-5 metacarpals 3–5 ph phalanx pi pisiform ul ulna ule ulnare un ungual. |
| Figure 67. Limbs of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Reconstructions based on an adult skeleton (SAM-PK-K1332). Right forearm, carpus and manus (A), right distal tarsals and pes (B), and right carpus (C) in dorsal view. Abbreviations: I-V digits I-V ce centrale dc1-5 distal carpals 1–5 dt3, 4 distal tarsal 3, 4 fo foramen mc1, 4, 5 metacarpal 1, 4, 5 mt1-4 metatarsals 1–4 ol olecranon ph phalanx pi pisiform ra radius rae radiale ul ulna ule ulnare un ungual. |
| Figure 68. Hip and posterior trunk of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Hip and posterior trunk in an adult skeleton (SAM-PK-K1332). Photograph (A) and line drawing (B) of the left ilium and pubis and sacral vertebrae in left lateral view. Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bar equals 3 cm. Abbreviations: ant antitrochanter CA caudal vertebra cat coossified anterior trochanter ch chevron D dorsal vertebra fe femur il ilium is ischium l left of obturator foramen ot ossified tendon poap postacetabular process pped pubic peduncle prap preacetabular process pu pubis r rib S sacral vertebra tp transverse process. |
| Figure 69. Hindlimb of Heterodontosaurus tucki from from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Tibiotarsus in an adult skeleton (SAM-PK-K1332). Photograph (A) and line drawing (B) of the left distal femur and tibiotarsus in lateral view. Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bar equals 5 cm. Abbreviations: at anterior trochanter ca calcaneum fe femur fi fibula ti tibia. |
| Figure 70. Hindlimb of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Tibiotarsus in an adult skeleton (SAM-PK-K1328). Photograph (A) and line drawing (B) of the right tibiotarsus in anterior view. Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bar equals 5 cm. Abbreviations: as astragalus asp ascending process at anterior trochanter ca calcaneum cc cnemial crest fi fibula ti tibia. |
| Figure 71. Pes of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Tarsometatarsus and pes of an adult skeleton (SAM-PK-K1328). Photograph (A) and line drawing (B) of the right tarsometatarsus and pes in anterior view. Hatching indicates broken bone; dashed lines indicate estimated edges; tone indicates matrix. Scale bar equals 3 cm. Abbreviations: I-IV digits I-IV ded dorsal extensor depression dip dorsal intercondylar process dt3, 4 distal tarsals 3, 4 fo foramen mt1-4 metatarsals 1-4 un ungual. |
| Figure 72. Skeleton of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Silhouette skeletal reconstruction in lateral view showing preserved bones (based on SAM-PK-K1332). Distal most caudal vertebrae unknown. |
| Figure 88. Hypotheses for jaw movement during tooth-to-tooth occlusion in Heterodontosaurus tucki. Jaw movements (arrows) to account for shearing wear facets during isognathous occlusion (from Crompton and Attridge 1986 [8]) A Dorsomedial movement of the lower jaws against stable maxillary tooth rows B Dorsal movement of the lower jaws with lateral flaring of the maxillary tooth rows C Medial rotation of the dentaries pivoting anteriorly at the predentary-dentary joint. |
| Figure 89. Occlusion and wear between maxillary and dentary teeth in Heterodontosaurus tucki. Reconstruction of left maxillary and dentary teeth in anterior view at different stages of wear A Dentition in a subadult with unworn crowns and near vertical occlusal plane with dashed lines indicating the occlusal surface of a worn crown (based on AMNH 24000) B Worn crowns from the middle of the tooth rows showing labiolingually concave wear facets, which are more concave in dentary than maxillary crowns (based on SAM-PK-K1332). The dentary tooth is shown at the initiation and conclusion of a masticatory stroke, with a dashed line showing the tooth margin hidden from view and registration dots and an arrow indicating tooth movement during occlusion. |
| Figure 90. Snout and upper dentition in Heterodontosaurus tucki. Cast of snout and upper dentition (UCRC PVC11) from an early mold of adult skull SAM-PK-K1332 A Cast of snout and upper dentition in left lateral view B Stereopair of anterior portion of the left antorbital fossa in posterolateral view. Scale bar equals 2 cm in A and 1 cm in B. Abbreviations: aantfe accessory antorbital fenestra eantfe external antorbital fenestra iantfe internal antorbital fenestra j jugal l lacrimal or left m maxilla m2, 10, maxillary tooth 2, 10 n nasal nf narial fossa pm premaxilla pm2, 3 premaxillary tooth 2, 3 pmfo promaxillary fossa r right. |
| Figure 91. Premaxillary dentition in Heterodontosaurus tucki. Cast of premaxillary tooth 2 and 3 in left lateral view (UCRC PVC11) from an early mold of adult skull SAM-PK-K1332. Stereopair (A) and line drawing (B) showing spalling, abrasion, and the mesial and distal edges of ligual tooth-to-bill wear facets. Scale bar equals 5 mm. Abbreviations: aa apical abrasion bsae breakage surface with abraded edges elwf edge of lingual wear facet sfs spalled fracture surface safs spalled abraded fracture surface. |
| Figure 92. Configuration of jaw articulation and long axis of the lower jaw in Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Diagrammatic depiction of jaw configuration based on subadult skull AMNH 24000 and adult skull SAM-PK-K1332 A Left quadrate condyles in anterior view showing 30° angle from the horizontal B Left quadrate condyles and long axis of the lower jaw in ventral view showing 40° angle from a transverse axis and 10° divergence from the midline, respectively. |
| Figure 93. Relative position of the lower jaw in Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Position of the lower jaw during occlusion (A) and moderate gape (B) as seen in left lateral view of a cast of the cranium and lower jaws of adult specimen SAM-PK-K1332. Scale bar equals 2 cm. |
| Figure 94. Previous jaw muscle reconstructions in Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa A Initial adductor muscle reconstruction (from Crompton and Attridge 1986 [8]) B Reconstruction of the adductor mandibulae externus superficialis (left) and the depressor mandibulae (right) (adapted from figures in Norman et al. 2011 [21]) C Reconstruction of the adductor mandibulae externus medialis (adapted from figures in Norman et al. 2011 [21]) D Reconstruction of the adductor mandibulae externus profundus (left) (misidentified as “adductor mandibulae externus posterior” in legend for Fig. 35, Norman et al. 2011 [21]) and the adductor mandibulae posterior (right) (adapted from figures in Norman et al. 2011 [21]) E Reconstruction of the pseudotemporalis (left) and the pterygoideus posterior (right) (adapted from figures in Norman et al. 2011 [21]). |
| Figure 95. Jaw musculature and keratin sheathing in Heterodontosaurus from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Reconstruction of select jaw muscles and keratin sheathing of upper and lower bills supported in this study. Pink tone indicates wear facets. Abbreviations: amep adductor mandibulae externus profundus ames adductor mandibulae externus superficialis amev adductor mandibulae externus ventralis dm depressor mandibulae. |
|
| The maximum number of images (100) is exceeded ! |
Other References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Crompton A, Charig A (1962) A new ornithischian from the Upper Triassic of South Africa. Nature 196: 1074-1077. doi: 10.1038/1961074a0
- ↑ Halstead T, Tarlo L, Tarlo B (1963) Convergent evolution in teeth. Royal Dental Hospital Magazine, London, n. s. 1: 18-19.
- ↑ Galton P (1970) Ornithischian dinosaurs and the origin of birds. Evolution 24: 448-462. doi: 10.2307/2406818
- ↑ 4.0 4.1 4.2 Santa Luca A, Crompton A, Charig A (1976) A complete skeleton of the Late Triassic ornithischian Heterodontosaurus tucki. Nature 264: 324-328.
- ↑ Thulborn R (1978) Aestivation among ornithopod dinosaurs of the African Trias. Lethaia 11: 185-198. doi: 10.1111/j.1502-3931.1978.tb01226.x
- ↑ 6.0 6.1 Hopson J (1980) Tooth function and replacement in early Mesozoic ornithischian dinosaurs: implications for aestivation. Lethaia 13: 93-105. doi: 10.1111/j.1502-3931.1980.tb01035.x
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 Weishampel D (1984) Evolution of jaw mechanisms in ornithopod dinosaurs. Advances in Anatomy and Cell Biology 87: 1-109. doi: 10.1007/978-3-642-69533-9_1
- ↑ 8.0 8.1 8.2 8.3 Crompton A, Attridge J (1986) Masticatory apparatus of the larger herbivores during Late Triassic and Early Jurassic times. In: Padian K (Ed). The Beginning of the Age of Dinosaurs: Faunal Change Across the Triassic-Jurassic Boundary. Cambridge University Press, London: 223-236.
- ↑ 9.0 9.1 Bakker R, Galton P (1974) Dinosaur monophyly and a new class of vertebrates. Nature 248: 168-172. doi: 10.1038/248168a0
- ↑ 10.0 10.1 10.2 Bakker R (1986) The Dinosaur Heresies. Bath Press, Avon, 481 pp.
- ↑ Galton P (1986) Herbivorous adaptations of Late Triassic and Early Jurassic dinosaurs. In: Padian K (Ed). The Beginning of the Age of Dinosaurs. Faunal Change Across the Triassic-Jurassic Boundary. Cambridge University Press, London and New York: 203-221.
- ↑ 12.0 12.1 Weishampel D, Witmer L (1990) Heterodontosauridae. In: Weishampel D Dodson P Osmólska H (Eds). The Dinosauria, first edition. University of California Press, Berkeley: 486-497.
- ↑ Norman D, Weishampel D (1991) Feeding mechanisms in some small herbivorous dinosaurs: processes and patterns. In: Rayner J Wootton R (Eds). , Biomechanics in Evolution. Cambridge University Press, Cambridge: 161-181.
- ↑ 14.0 14.1 Brett-Surman M (1997) Ornithopods. In: Farlow J Brett-Surman M (Eds). , The Complete Dinsoaur. Indiana Univeristy Press, Bloomington: 330-346.
- ↑ Norman D, Sues H, Witmer L, Coria R (2004) Basal Ornithopoda. In: Weishampel D Dodson P Osmólska H (Eds). The Dinosauria, 2nd ed. University of California Press, Berkeley: 393-412. doi: 10.1525/california/9780520242098.003.0021
- ↑ 16.0 16.1 Langer M, Benton M (2006) Early dinosaurs: a phylogenetic study. Journal of Systematic Palaeontology 4: 1-50. doi: 10.1017/S1477201906001970
- ↑ Butler R, Porro L, Norman D (2008) A juvenile skull of the primitive ornithischian dinosaur Heterodontosaurus tucki from the ‘Stormberg’ of southern Africa. Journal of Vertebrate Paleontology 28: 702-711. doi: [702:AJSOTP2.0.CO;2 10.1671/0272-4634(2008)28[702:AJSOTP]2.0.CO;2]
- ↑ Smith J (1997) Heterodontosauridae. In: Currie P Padian K (Eds). Encyclopedia of Dinosaurs. Academic Press, San Diego: 317-320.
- ↑ Brusatte S, Nesbitt S, Irmis R, Butler R, Benton M, Norell M (2010) The origin and early evolution of dinosaurs. Earth-Science Reviews, London, 101: 68-100. doi: 10.1016/j.earscirev.2010.04.001
- ↑ 20.0 20.1 Porro L, Butler R, Barrett P, Moore-Fay S, Abel R (2011) New heterodontosaurids specimens from the Lower Jurassic of southern Africa and the early ornithischian dinosaur radiation. Earth and Environmental Transactions of the Royal Society of Edinburgh 101: 351-366. doi: 10.1017/S175569101102010X
- ↑ 21.00 21.01 21.02 21.03 21.04 21.05 21.06 21.07 21.08 21.09 21.10 21.11 21.12 21.13 21.14 21.15 21.16 21.17 21.18 21.19 21.20 21.21 21.22 21.23 21.24 21.25 21.26 21.27 21.28 21.29 21.30 21.31 21.32 21.33 21.34 21.35 21.36 21.37 21.38 Norman D, Crompton A, Butler R, Porro L, Charig A (2011) The Lower Jurassic ornithischian dinosaur Heterodontosaurus tucki Crompton and Charig 1962: cranial anatomy, functional morphology, taxonomy, and relationships. Zoological Journal of the Linnean Society 162: 182-279.
- ↑ Kitching J, Raath M (1984) Fossils from the Elliot and Clarens Formations (Karoo Sequence) of the northeastern Cape, Orange Free State and Lesotho, and a suggested biozonation based on tetrapods. Palaeontologia africana 25: 111-125.
- ↑ Smith R (1990) A review of stratigraphy and sedimentary environments of the Karoo Basin of South Africa. Journal of African Earth Sciences (and the Middle East) 10: 117-137. doi: 10.1016/0899-5362(90)90050-O
- ↑ Knoll F (2005) The tetrapod fauna of the Upper Elliot and Clarens formations in the main Karoo Basin (South Africa and Lesotho). Bulletin de la Société géologique de France 176: 81-91. doi: 10.2113/176.1.81
- ↑ Gradstein F, Ogg J (2009) The geologic time scale. In: Hedges S Kumar S (Eds). , The Timetree of Life. Oxford University Press, Oxford: 26-34.
- ↑ Steel R (1969) Ornithischia. Handbuch der Paläoherpetologie 15: 1-84.
- ↑ 27.0 27.1 27.2 Pol D, Rauhut O, Becerra M (2011) A Middle Jurassic heterodontosaurid dinosaur from Patagonia and the evolution of heterodontosaurids. Naturwissenschaften 98: 369-379.
- ↑ 28.0 28.1 28.2 28.3 28.4 28.5 Santa Luca A (1980) The postcranial skeleton of Heterodontosaurus tucki (Reptilia, Ornithischia) from the Stormberg of South Africa. Annals of the South African Museum 79: 159–211. doi: 10.1038/264324a0
- ↑ Thulborn R (1974) A new heterodontosaurid dinosaur (Reptilia: Ornithischia) from the Upper Triassic Red Beds of Lesotho. Zoological Journal of the Linnean Society 55: 151-175. doi: 10.1111/j.1096-3642.1974.tb01591.x
- ↑ 30.0 30.1 30.2 Charig A, Crompton A (1974) The alleged synonymy of Lycorhinus and Heterodontosaurus. Annals of the South African Museum 64: 167-189.
- ↑ Witmer L, Sampson S, Solounias N (1999) The proboscis of tapirs (Mammalia: Perissodactyla): a case study in novel narial anatomy. Journal of Zoology London 249: 249-267. doi: 10.1111/j.1469-7998.1999.tb00763.x
- ↑ 32.0 32.1 Witmer L (1997) The evolution of the antorbital cavity of archosaurs: A study in soft-tissue reconstruction in the fossil record with an analysis of the function of pneumaticity. Journal of Vertebrate Paleontology, Memoir 3 17: 1–73.
- ↑ 33.0 33.1 33.2 33.3 33.4 Martinez R, Sereno P, Alcober O, Colombi C, Renne PR et a (2011) A basal dinosaur from the dawn of the dinosaur era in southwestern Pangaea. Science 331: 206-210. doi: 10.1126/science.1198467
- ↑ Peng G (1992) Jurassic ornithopod Agilisaurus louderbacki (Ornithopoda: Fabrosauridae) from Zigong, Sichuan, China. Vertebrata Palasiatica 30: 39-51.
- ↑ 35.0 35.1 35.2 35.3 35.4 35.5 35.6 35.7 35.8 Sereno P (1991) Lesothosaurus, “fabrosaurids,” and the early evolution of Ornithischia. Journal of Vertebrate Paleontology 11: 168–197. doi 10.1080/02724634.1991.10011386
- ↑ 36.0 36.1 36.2 36.3 36.4 36.5 Galton P (1974a) The ornithischian dinosaur Hypsilophodon from the Wealden of the Isle of Wight. Bulletin, British Museum (Natural History) Geology 25: 1-152.
- ↑ Butler R (2005) The ‘fabrosaurid’ ornithischian dinosaurs of the Upper Elliot Formation (Lower Jurassic) of South Africa and Lesotho. Zoological Journal of the Linnean Society 145: 175-218. doi: 10.1111/j.1096-3642.2005.00182.x
- ↑ Galton P (1974b) Notes on Thescelosaurus, a conservative ornithopod dinosaur from the Upper Cretaceous of North America, with comments on ornithopod classification. Journal of Paleontology 48: 1048-1067.
- ↑ Sereno P (2009) Comparative cladistics. Cladistics 25: 624-659. doi: 10.1111/j.1096-0031.2009.00265.x
- ↑ 40.0 40.1 40.2 Sereno P (2007) Basal Sauropodomorpha: historical and recent phylogenetic hypotheses, with comments on Ammosaurus major (Marsh, 1889). In: Barrett P Batten D (Eds). Evolution and Paleobiology of Early Sauropodomorph Dinosaurs. Special Papers in Palaeontology 77. The Palaeontological Association, London: 261-289.
- ↑ 41.0 41.1 Sereno P (1993) The pectoral girdle and forelimb of the basal theropod Herrerasaurus ischigualastensis. Journal of Vertebrate Paleontology 13: 425-450. doi: 10.1080/02724634.1994.10011524
- ↑ Owen R (1861) Monograph on the fossil Reptilia of the Wealden and Purbeck Formations. Part V. Lacertilia. Palaeontographical Society Monographs 12: 31-39.
- ↑ Broom R (1911) On the dinosaurs of the Stormberg, South Africa. Annals of the South African Museum 7: 291-307.
- ↑ Butler R, Galton P, Porro L, Chiappe L, Henderson DM et a (2010) Lower limits of ornithischian dinosaur body size inferred from a new Upper Jurassic heterodontosaurid from North America. Proceedings of the Royal Society B: Biological Sciences 277: 375-381. doi: 10.1098/rspb.2009.1494
- ↑ Zheng X, You H, Xu X, Dong Z (2009) Early Cretaceous heterodontosaurid dinosaur with integumentary structures. Nature 458: 333–336. doi: 10.1038/nature07856
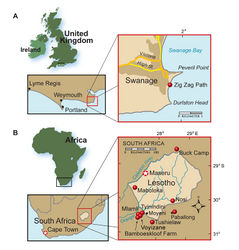
![Figure 2. Early heterodontosaurid discoveries. A Lithographic drawing of the right and left premaxillae and the anterior portion of the left maxilla in lateral view of Echinodon becklesii (NHMUK 48209; from Owen 1861[42]) B Drawing of lower jaws in dorsal view of Geranosaurus atavus (SAM-PK-K1871; from Broom 1911[43]) C Photograph of the posterior portion of a subadult skull in right lateral view of Heterodontosaurus tucki (AMNH 24000). Scale bars equal 1 cm in A and 2 cm in B and C.](https://species-id.net/o/thumb.php?f=ZooKeys-226-001-g002.jpg&width=175)
![Figure 9. More recent heterodontosaurid discoveries from northern locales. A Jaws of Fruitadens haagarorum from the Upper Jurassic Morrison Formation in Colorado, USA (based on LACM 115747, 128258; reversed from Butler et al. 2010[44]) B Left dentary in lateral view of an undescribed heterodontosaurid from the Lower Jurassic Kayenta Formation of Arizona (from Sereno et al. unpublished) C Partial skull of Tianyulong confuciusi from the Yixian Formation of Liaoning Province, PRC (STMN 26-3; reversed from Zheng et al. 2009[45]). Abbreviations: a angular ad 9, 10 alveolus for dentary tooth 9, 10 adf anterior dentary foramen antfo antorbital fossa apd articular surface for the predentary d dentary d1, 2, 8 dentary tooth 1, 2, 8 emf external mandibular fenestra en external nares j jugal l lacrimal m maxilla n nasal pd predentary pf prefrontal pm premaxilla po postorbital q quadrate qj quadratojugal sa surangular. Scale bar equals 1 cm in A and C and 5 mm in B.](https://species-id.net/o/thumb.php?f=ZooKeys-226-001-g009.jpg&width=201)

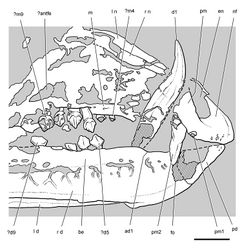
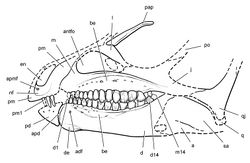
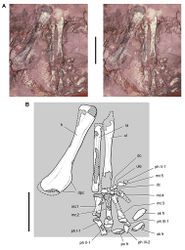
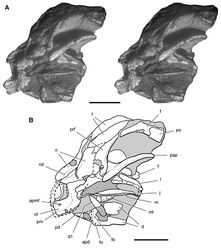
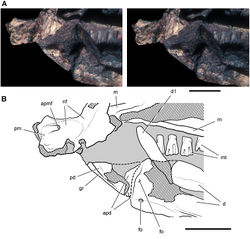

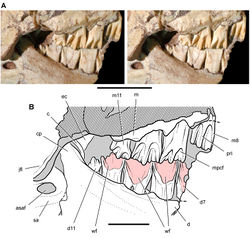
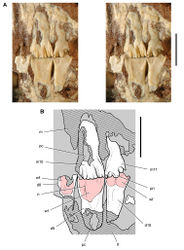
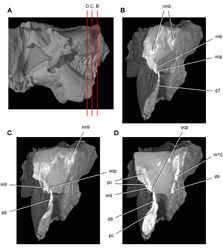
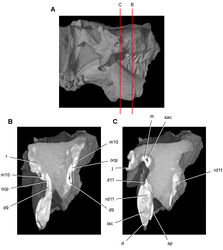
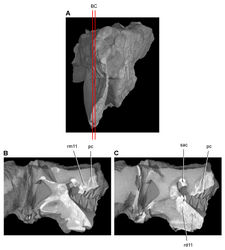

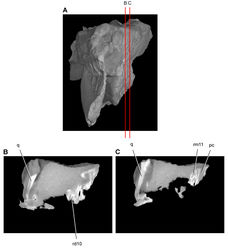
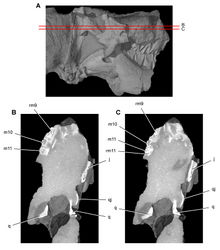
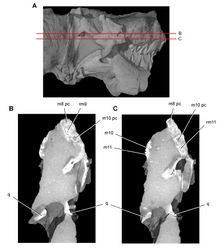



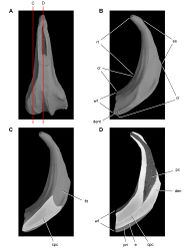
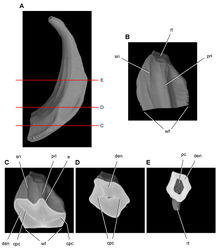
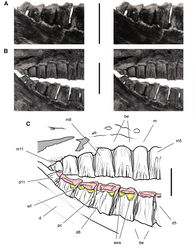
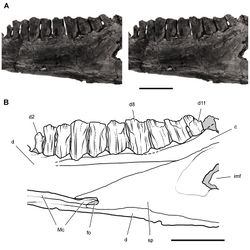
![Figure 57. Skull of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Previous skull reconstructions in left lateral view A From Charig and Crompton (1974)[30] B Reversed from Weishampel (1984)[7].](https://species-id.net/o/thumb.php?f=ZooKeys-226-001-g057.jpg&width=245)
![Figure 58. Skull of Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa. Skull reconstruction in left lateral view from Norman et al. (2011)[21]. Abbreviations: a angular antfe antorbital fenestra antfo antorbital fossa ar articular bo basioccipital c coronoid d dentary f frontal j jugal jfl jugal flange jh jugal horn l lacrimal m maxilla n nasal nf narial fossa p parietal pap palpebral pd predentary pm premaxilla po postorbital popr paroccipital process prf prefrontal q quadrate qf quadrate foramen qj quadratojugal sa surangular sq squamosal.](https://species-id.net/o/thumb.php?f=ZooKeys-226-001-g058.jpg&width=250)
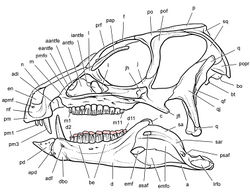

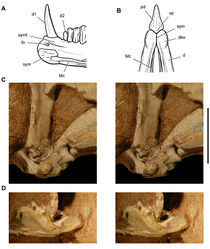
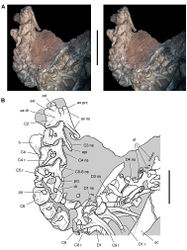
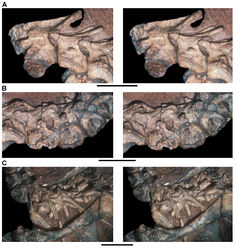
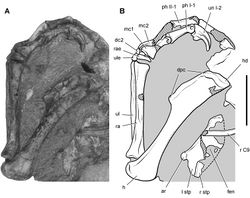
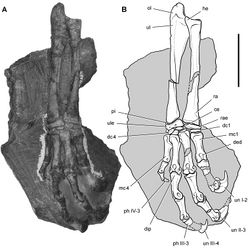
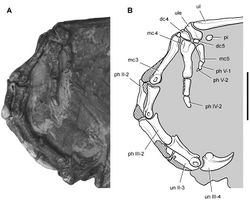
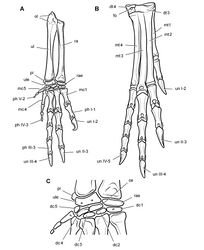
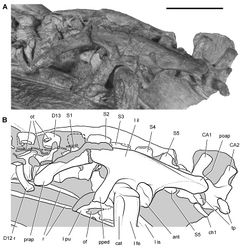
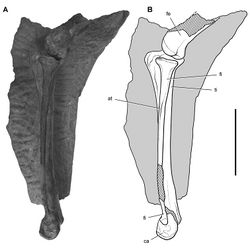
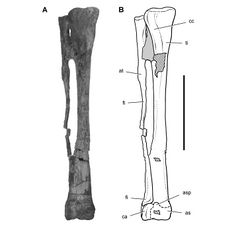
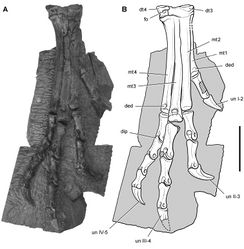
![Figure 88. Hypotheses for jaw movement during tooth-to-tooth occlusion in Heterodontosaurus tucki. Jaw movements (arrows) to account for shearing wear facets during isognathous occlusion (from Crompton and Attridge 1986[8]) A Dorsomedial movement of the lower jaws against stable maxillary tooth rows B Dorsal movement of the lower jaws with lateral flaring of the maxillary tooth rows C Medial rotation of the dentaries pivoting anteriorly at the predentary-dentary joint.](https://species-id.net/o/thumb.php?f=ZooKeys-226-001-g088.jpg&width=235)
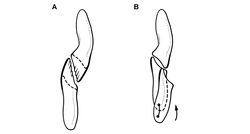
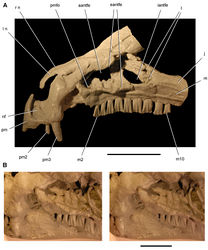

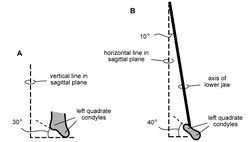
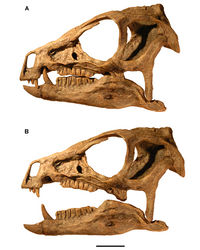
![Figure 94. Previous jaw muscle reconstructions in Heterodontosaurus tucki from the Lower Jurassic Upper Elliot and Clarens formations of South Africa A Initial adductor muscle reconstruction (from Crompton and Attridge 1986[8]) B Reconstruction of the adductor mandibulae externus superficialis (left) and the depressor mandibulae (right) (adapted from figures in Norman et al. 2011[21]) C Reconstruction of the adductor mandibulae externus medialis (adapted from figures in Norman et al. 2011[21]) D Reconstruction of the adductor mandibulae externus profundus (left) (misidentified as “adductor mandibulae externus posterior” in legend for Fig. 35, Norman et al. 2011[21]) and the adductor mandibulae posterior (right) (adapted from figures in Norman et al. 2011[21]) E Reconstruction of the pseudotemporalis (left) and the pterygoideus posterior (right) (adapted from figures in Norman et al. 2011[21]).](https://species-id.net/o/thumb.php?f=ZooKeys-226-001-g094.jpg&width=220)