Ablattaria subtriangula
| Notice: | This page is derived from the original publication listed below, whose author(s) should always be credited. Further contributors may edit and improve the content of this page and, consequently, need to be credited as well (see page history). Any assessment of factual correctness requires a careful review of the original article as well as of subsequent contributions.
If you are uncertain whether your planned contribution is correct or not, we suggest that you use the associated discussion page instead of editing the page directly. This page should be cited as follows (rationale):
Citation formats to copy and paste
BibTeX: @article{Qubaiová2015ZooKeys, RIS/ Endnote: TY - JOUR Wikipedia/ Citizendium: <ref name="Qubaiová2015ZooKeys">{{Citation See also the citation download page at the journal. |
Ordo: Coleoptera
Familia: Silphidae
Genus: Ablattaria
Name
Ablattaria subtriangula Reitter, 1905 – Wikispecies link – Pensoft Profile
Type material examined
Lectotype male (here designated) (MNHN, coll. Pic), pinned, labelled: “SIERRA DE GUADALUPE / (CÁCERES) / MAI 1904 / G. SCHRAMM [p] // 71 [hw] // type [hw] // Ablattaria / laevigata / v. subtriangula Rtt. / (Reitt. vid.) [hw, probably Portevin’s handwriting] // Ablattaria subtriangula / Reitt. [hw, Pic’s handwriting] // Lectotype / Ablattaria subtriangula / Reitter, 1905 / J. Qubaiová & J. Růžička / des. 2014 [p, red label]. Paralectotype male (MNHN, coll. Marmottan) (Fig. 27), pinned, labelled: “SIERRA DE GUADALUPE / (CÁCERES) / MAI 1904 / G. SCHRAMM [p] // TYPE [p, red modern label] // Subtriangula / Reitt. / (Reynoza) [hw] // Muséum Paris / 1914 / Coll. H. Marmottan [p, modern label] // Paralectotype / Ablattaria subtriangula / Reitter, 1905 / J. Qubaiová & J. Růžička des. 2014 [p, red label]”.
Additional material examined
18 specimens, see Appendix 1.
Diagnostic description
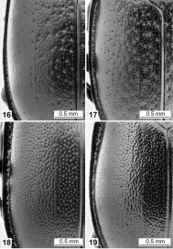
Total body length 12–16 mm, body glossy, black (Figs 3, 14, 19). Pronotum almost conical in shape, margins anterolaterally constricted (Fig. 14); with evenly distributed, homogenous, distinct punctures on whole dorsal surface (Fig. 14). Elytra regularly rounded; with densely and evenly distributed medium-sized, distinct punctures of similar size (Fig. 19).
Remarks
Additional male specimen (MNHN, coll. Marmottan), pinned, labelled: “Soto [hw] // TYPE [p, red modern label] // Silpha subtriangula / Reitt. / Co-type [hw, same handwriting as on identification label of lectotype specimen]” is not considered here as paralectotype, because its locality is not consistent with precise information provided in the original description by Reitter (1905)[1]. “Soto” is vague, as there seem to be more than 10 localities with this name across Spain (http://en.wikipedia.org/wiki/Soto), none of which are in either Cáceres Province or elsewhere in the Extramadura autonomous community.
Biology
Regarding the seasonal activity of Ablattaria subtriangula, in the limited adult material examined, most specimens were collected between April and June.
Distribution
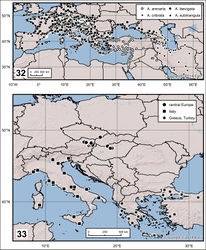
Endemic to continental Spain (Fig. 32).
Geometric morphometrics Relative warps (RWs) of both males and females of the four Ablattaria taxa were calculated and plotted on an axis system. The first RW (RW1) axis represented 44.25% of shape variability and the second axis (RW2) accounted for 20.22%. Subsequently, discriminant analysis (DA) was applied between the sexes on the first 30 axes representing 99.94% of variability. The results indicated shape sexual dimorphism (Hotelling’s test: 444.2, F: 14.071, p < 0.0001). Specimens correctly classified to their means showed a percentage of 82.59.
Male groups of the four taxa were tested independently and RW1 accounted for 44.74% of the total variance whereas RW2 accounted for 20.43%. A higher 46.98% of variability was explained by the RW1 axis in females and 18.73% by RW2. Both scatter plots of the two first RWs for male and female Ablattaria displayed a high overlap between the groups of the different taxa. The thin-plate spline (TPS) transformation grids (not included in the article) indicated some shape differences between the taxa especially in Ablattaria arenaria; less rounded or curved pronotal margins posteriorly and more parallel elytra. In Ablattaria laevigata the posterior pronotal margins appeared more rounded (semi-elliptical) and the elytra were more robust than the other taxa particularly in the females, whereas the pronotal shape of Ablattaria subtriangula was more narrowed to the front (conical).
Multivariate analysis of variance (MANOVA) was performed on the four groups. The results indicated significant shape variations, but the separations between the groups were weak, given that the number of Ablattaria subtriangula specimens was very low compared to those of other groups. Hence, the analysis was repeated without the Ablattaria subtriangula samples to obtain a clearer separation.
Shape diversity of both pronotum and elytra between the three taxa was indicated by MANOVA. Male groups revealed significant shape differences (F = 32.93; Wilk’s lambda = 0.0784; DF = 40/512; p < 0.00001). Female groups demonstrated higher body shape variability (F = 24.93; Wilk’s lambda = 0.1252; DF = 40/546; p < 0.00001).
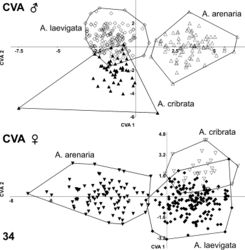
Two individual canonical variate analyses (CVA) for males and females (separately) were performed to obtain separation of the four groups on the first 20 axes of the RW scores matrix. These axes covered 99.81% of the shape variation between male groups and 99.82% between female groups. Results indicated no overlap between Ablattaria arenaria and either Ablattaria laevigata or Ablattaria cribrata in males and only with one specimen in females (Fig. 34). The overlap between Ablattaria laevigata and Ablattaria cribrata was minimal and more evident in males than in females. The jackknifed (or leave-one-out) values of the confusion matrix in Ablattaria laevigata males illustrated a correct mean classification of 131 from 144 specimens (13 showed means closer to that of Ablattaria cribrata). In Ablattaria arenaria, this was the case for 84 of 85 (1 was closer in its mean value to that of Ablattaria cribrata). Ablattaria cribrata had 42 accurate classifications of 49 in total (7 specimens were closer to Ablattaria laevigata). In the females, 150 specimens of 175 in Ablattaria laevigata were correctly classified (24 were closer to Ablattaria cribrata and 1 to Ablattaria arenaria). In Ablattaria arenaria, 82 of 87 were correctly classified (4 were closer to Ablattaria laevigata and 1 to Ablattaria cribrata). In Ablattaria cribrata, 25 of 33 were clearly classified (8 were closer to Ablattaria laevigata). These findings strongly indicate the shape variations of these taxa, and thus support the hypothesis that all three taxa constitute separate species.
Nevertheless, both males and females (independently) of Ablattaria subtriangula were tested and compared with one group formed by the three other taxa to ensure its independence by discriminant analysis (DA). Results indicated significant shape variability in males (Hotelling’s test: 20.598, F: 5.0946, p < 0.001) with 86.93% correct classification of specimens to their means. For females (Hotelling’s test: 40.282, F: 10.465, p < 0.0001), specimens correctly classified were 85.48%. As a result, Ablattaria subtriangula indicates its division from the other taxa and therefore may also be considered as a separate species.
To examine allometry effects, the influence of size on body shape was tested first on the four taxa by separating them into groups based on taxon and sex. The multivariate regressions of shape onto size were performed one group at a time. Results showed significant relationship in both sexes of Ablattaria laevigata, males of Ablattaria arenaria and Ablattaria subtriangula, and females of Ablattaria cribrata. The results were insignificant for female Ablattaria arenaria, Ablattaria subtriangula and male Ablattaria cribrata (Table 2).
| Species | Ablattaria laevigata | Ablattaria arenaria | ||||
|---|---|---|---|---|---|---|
| Explained variance | Goodall’s F-test | Explained variance | Goodall’s F-test | |||
| Sex | F-value | p-value | F-value | p-value | ||
| Males | 1.61% | 2.33 | p < 0.0001 | 1.70% | 1.45 | p < 0.01 |
| Females | 0.91% | 1.59 | p < 0.0001 | 1.60% | 1.34 | p < 0.1 |
| Species | Ablattaria cribrata | Ablattaria subtriangula | ||||
| Explained variance | Goodall’s F-test | Explained variance | Goodall’s F-test | |||
| Sex | F-value | p-value | F-value | p-value | ||
| Males | 1.30% | 0.61 | p = 0.9994 | 51.03% | 3.13 | p < 0.0001 |
| Females | 7.60% | 2.55 | p < 0.0001 | 4.79% | 0.3 | p = 1.00 |
| Explained variance | Goodall’s F-test | ||
|---|---|---|---|
| Sex | F-value | p-value | |
| Males | 16.09% | 6.57 | p < 0.00001 |
| Females | 11.14% | 4.61 | p < 0.00001 |
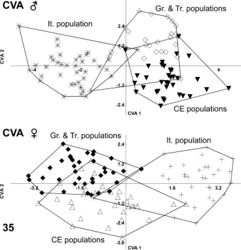
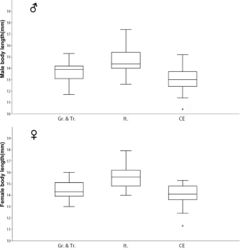
MANOVA was performed subsequently. Male populations revealed significant shape dissimilarity (F = 10.35; Wilk’s lambda = 0.121; DF = 30/166; p < 0.00001). Shape variability was found to be also significant in the female populations (F = 8.337; Wilk’s lambda = 0.166; DF = 30/172; p < 0.00001). Canonical variate analysis on the first 15 axes was performed and represented 99.54% of the shape variation in males and 99.68% in females. Results indicated overlap between all groups (Fig. 35). The jackknifed values of the confusion matrix for both sexes are presented in Tables 4 and 5. The most obvious separation was seen in the Italian population, which showed incorrect classification of only 7 specimens in the two sexes taken together of a total 72 specimens. The Central European population showed higher variation in the male than was that in the female populations from Greece and Turkey.
| Ablattaria laevigata populations, males | Pop. (Gr. & Tr.) | Pop. (It.) | Pop. (CE) | Total |
|---|---|---|---|---|
| Pop. (Gr. & Tr.) | 15 | 4 | 7 | 26 |
| Pop. (It.) | 3 | 35 | 1 | 39 |
| Pop. (CE) | 7 | 0 | 28 | 35 |
| Ablattaria laevigata populations, females | Pop. (Gr. & Tr.) | Pop. (It.) | Pop. (CE) | Total |
|---|---|---|---|---|
| Pop. (Gr. & Tr.) | 27 | 1 | 9 | 37 |
| Pop. (It.) | 0 | 30 | 3 | 33 |
| Pop. (CE) | 10 | 2 | 21 | 33 |
| Explained variance | Goodall’s F-test | |||
|---|---|---|---|---|
| Sex | F-value | DF | p-value | |
| Males | 11.49% | 4.16 | 318/10176 | p < 0.00001 |
| Females | 9.91% | 3.63 | 318/10494 | p < 0.00001 |
Taxon Treatment
- Qubaiová, J; Růžička, J; Šípková, H; 2015: Taxonomic revision of genus Ablattaria Reitter (Coleoptera, Silphidae) using geometric morphometrics ZooKeys, (477): 79-142. doi
Images
|